Question: Using resonance structures of the intermediates, explain why bromination of biphenyl occurs at ortho and Para positions rather than at Meta. Biphenyl
Using resonance structures of the intermediates, explain why bromination of biphenyl occurs at ortho and Para positions rather than at Meta.
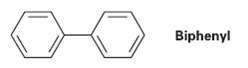
Biphenyl
Step by Step Solution
3.40 Rating (169 Votes )
There are 3 Steps involved in it
Resonance structures explain that bromination occurs in the ortho and Para pos... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-B (43).docx
120 KBs Word File


