What is the hybridization at each nitrogen of the amino acid histidine? What kind of orbital is
Question:
What is the hybridization at each nitrogen of the amino acid histidine? What kind of orbital is occupied by the unshared pair of electrons on each nitrogen? Explain.
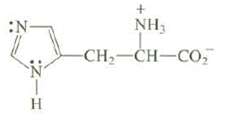
Transcribed Image Text:
:N :Z-H H + NH₂ CH, CH–CO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Nitrogen 1 is sp hybridized and its unshared pair of electrons i...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
What is the hybridization at all atoms, except hydrogen's in these compounds? a) CHNH, d) b) CH=CHCHC=N OH 6 H NH
-
A dipole of 6.6 nC point charges arranged 0.16 cm apart is centered at the origin and oriented along the x-axis. The positive charge is on the negative x axis. How m energy is required to place a...
-
Broome Instruments Company manufactures a control valve used in air-conditioning systems. The firm uses a standard costing system for product costing. The manufacturing overhead rate is based on a...
-
1. Explain the relationship between Coca-Colas objective, decision, and problem in this case. 2. What is the classification of the problem/opportunity and which decision-making model should have been...
-
What is the minimum water depth necessary for a 40 -ftwide stream to handle \(4000 \mathrm{ft}^{3} / \mathrm{s}\) if the flow is not supercritical?
-
Phillips, Inc., a cash basis C corporation, completes $100,000 in sales for year 1, but only $75,000 of this amount is collected during year 1. The remaining $25,000 from these sales is collected...
-
Hi - Low Ltd has analysed its distribution costs and activity over the past 3 months and these are as follows: Monthly Distribution costs Units sold January 1 6 1 , 0 0 0 5 0 , 0 0 0 February 2 5...
-
Childrens Best Hope (CBH) provides day care ser-vices to low income families. CBH bills the state for its services under a service contract. Billings for the first four months of 2013 are anticipated...
-
Show a MO energy level diagram for the neutral molecule HeH. Use this diagram to explain whether HeH is expected to stable or not?
-
At a pH of 10.8, the amino acid arginine exists primarily as the following dipolar ion. Show the resonance structures for the cationic part of arginine and discuss their relative contributions to the...
-
The line y = mx + 6 is a tangent to the curve y = x 2 - 4x + 7. Find the possible values of m.
-
Blair Company has $\$ 5$ million in total assets. The company's assets are financed with $\$ 1$ million of debt and $\$ 4$ million of common equity. The company's income statement is summarized...
-
An advanced engineering computer was purchased for $\$ 22,750$ and its expected life is 4 years. It is estimated the company could sell the computer for $\$ 500$ after 5 years. Compute and compare...
-
Explain the difference between Inverse ETFs and Leverage ETFs.
-
Your colleague is excited about your good fortune (Problem 3.1) at work, but she only got the promise of a watch or \($300\) cash. You convince her that she will be better in the long run by just...
-
Suppose you make 15 equal annual deposits of \($1,000\) each into a bank account paying 5% interest per year. The first deposit will be made one year from today. How much money can be withdrawn from...
-
The following data give real GDP, Y, capital, \(K\), and labor, \(N\), for the U.S. economy in various years. Units and sources are the same as in Table 3.1. Assume that the production function is...
-
True & False The basis of an asset must be reduced by the depreciation allowable, 2. Adjusted gross income (AGI) is the basis for a number of phase-outs of deductions. 3. A change to adjusted gross...
-
Test the series for convergence or divergence. (-1)+1, M=1 n? 3 n' + 1
-
Draw the two chair conformations of menthol, and tell which is morestable. C Menthol CH(CH3)2
-
There are four cistransisomers of menthol (Problem 4.37), including the one shown. Draw the other three.
-
Identify each pair of relationships among the ? OH groups in glucose (red?blue, red?green, red?black, blue?green, blue?black, green?black) as cis or trans. CH2OH OH OH Glucose OH
-
What are the 2 main dimensions of an organizational culture we must consider when we initiate a lean deployment? Briefly explain each dimension
-
Mianzi, or face, is an important concept in Chinese negotiations and the wider environment of Chinese business culture. What does this concept mean?
-
Homestead Crafts, a distributor of handmade gifts, operates out of owner Emma Finn's house. At the end of the current period, Emma looks over her inventory and finds that she has 1,400 units...

Study smarter with the SolutionInn App


