Which of the following molecules has a dipole moment? Indicate the expected direction ofeach. (b) (d) (c)
Question:
Which of the following molecules has a dipole moment? Indicate the expected direction ofeach.
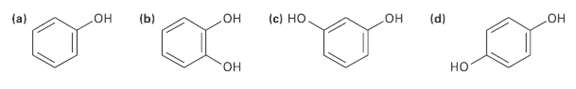
Transcribed Image Text:
(b) (d) (c) Но Он Он Он (a) но Он но
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a b CI ...View the full answer

Answered By

User L
I am a proficient writer with vast experience in medicine, nursing, biochemistry, microbiology, biology chemistry, accounting, economics, mathematics, statistics, actuarial science, psychology, sociology, philosophy, and social science. I deliver papers which are original, free from plagiarism, free from grammatical errors and high-quality in time
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following molecules has the largest a value: CH4, F2, C6H6, Ne?
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
The PF3 molecule has a dipole moment of 1.03 D, but BF3 has a dipole moment of zero. How can you explain the difference?
-
When a cosmetic manufacturer tests the market to determine how many women will buy eyeliner that has been tested for safety without subjecting animals to injury, is it involved in a descriptive...
-
Tyneka inherited 1,000 shares of Aqua, Inc. stock from Joe. Joe's basis was $35,000, and the fair market value on July 1, 2016 (the date of death) was $45,000. The shares were distributed to Tyneka...
-
Explain why colligative properties depend only on the concentration, and not on the identity of the molecule.
-
For each of the following sets of numbers, calculate a \(95 \%\) confidence interval for the mean ( \(\sigma\) not known); before going through the steps in calculating the confidence interval, the...
-
Demand for rug- cleaning machines at Clyde's U- Rent- It is shown in the following table. Machines are rented by the day only. Profit on the rug cleaners is $ 10 per day. Clyde has four rug-cleaning...
-
how does "coffee culture" vary around the world? Do you think Starbucks will succeed in Italy? Why do entrepreneurs in many parts of the world open coffee shops? What accounts for the popularity of...
-
Nine months ago, Muriel agreed to pay Aisha $1200 and $800 on dates 6 and 12 months, respectively, from the date of the agreement. With each payment Muriel agreed to pay interest at the rate of 8 %...
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
(a) The H C1 bond length is 136 pm. What would the dipole moment of HC1 be if the molecule were 100% ionic, H+ C1-? (b) The actual; dipole moment HC1 is 1.08 D. What is the percent ionic character...
-
In solving a linear program, no feasible solution exists. To resolve this problem we might a. add another variable. b. add another constraint. c. remove or relax a constraint. d. try a different...
-
The current zero-coupon yield curve for risk-free bonds is as follows: Maturity (years) 5 5 YTM 4.96% 5.53% 5.78% 5.98% 6.04% What is the price per $100 face value of a four-year, zero-coupon,...
-
What are the implications of cryptic diversity, species crypticism, and species complexes for biodiversity assessment, conservation prioritization, and ecosystem management, particularly in cryptic...
-
Using UK law, define the civil law and criminal law, compare civil law with criminal law and analyse the impact of English civil law and criminal law on business and professional services.
-
Lion I Of 1 0 of 10 points Question 1 Unlimited tries At one college, the tuition for a full-time student is $8,000 per semester. It has been announced that the tuition will increase by 3 percent...
-
WHAT IS SUBSTANTIVE CRIMINAL LAW VERSUS PROCEDURAL CRIMINAL LAW ?
-
Several college freshmen enter the deans private office and remain there for several hours. They refuse to let the dean leave until he yields to their demands to allow unrestricted visitation in all...
-
Imagine that your best friend knows you are taking a psychology course and wonders what psychology is all about. How would you define psychology for your friend? Write an essay on the discipline of...
-
A gas is continuously passed through an adiabatic turbine at the rate of 2 mol/s. Its initial temperature is 600 K, its initial pressure is 5 bar and its exiting pressure is 1 bar. Determine the...
-
Bromine is larger than chlorine, yet the two atoms have identical axial destabilization energies. Explain.
-
Draw the stereo isomers of these compounds: (a) 1, 3-Dimethyleyclohexane (b) 1, 2-Diethylcycloproane (c) 1-Chloro-3-methylcyclopentane
-
Draw both chair conformations of trans-1, 3-dimethyl cyclohexane indicate whether each methyl group is axial or equatorial.
-
You will modify the add method in the LinkedBag class. The add method is the only thing you should modify in LinkedBag. Add a second parameter to the method header that will be a boolean variable:...
-
Generate Random Decals is primitive please fix that as well As a license plate number, integers are to be used. Positive numbers add a car, negative numbers remove a car, zero stops the...
-
Describe the competitive positions in the drone industry and assess the opportunities for at least three positions in the industry.

Study smarter with the SolutionInn App


