Which of these compounds will have more of the conformation with the substituent on the cyclohexane ring
Question:
Which of these compounds will have more of the conformation with the substituent on the cyclohexane ring axial present at equilibrium?
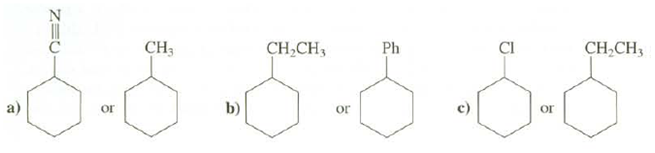
Transcribed Image Text:
CH,CH3 CI CH3 Ph CH,CH, b) or or or
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
Substituents larger than H prefer to be equatorial on a cyclohexane ring Larger substituents have a ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist? a) CHCHCH=CHCH CH3 c) CHC=CHCH b) CH3CHCH=CH CI d) CHC=CHCHCH3
-
Which of these compounds can form an intra molecular (within the same molecule) hydrogen bond between the hydrogen of the carboxylic acid and the oxygen of the ether group? Explain. H, H, H -C-c-c- |
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Find f. f(x) = f'(x) = 12x + X x > 0, f(1) = -4
-
Kevin Ltd is developing departmental overhead rates based on machine hours for its Moulding Department and direct labour hours for its Assembly Department. The Moulding Department has 20 machines,...
-
Why does corporate governance influence company value?
-
How can we use packaging to structure modeling artifacts?
-
Acme Corporation (a U.S. company located in Sarasota, Florida) has the following import/export transactions in 2011: March 1 Bought inventory costing 50,000 pesos on credit. May 1 Sold 60 percent of...
-
Golden Paper Company purchases a 90-day negotiable CD with a $1 million denomination from Vail National Bank, bearing a 3.25% annual yield. How much will Golden Paper receive from Vail when the CD...
-
What is the reduced form of the function F = X(X+Y)+ X(X+Y)? ) Y b) ) 0 d) 1
-
Draw the two chair conformations for ethyl cyclohexane which is more stable.
-
Bromine is larger than chlorine, yet the two atoms have identical axial destabilization energies. Explain.
-
Brooks Corp. is a medium-sized corporation specializing in quarrying stone for building construction. The company has long dominated the market, at one time achieving a 70% market penetration. During...
-
The Government Accountability Office Report Regulatory Coverage Generally Exists for Financial Planners, but Consumer Protection Issues Remain provides the most current overview licensing...
-
What are the various forms of virtual communication used in modern organizations?
-
How does one choose between communication methods and handle barriers to effective communication?
-
What are the issues surrounding smartphones, social media, and cybersecurity confronting modern organizations?
-
What are the types of interpersonal communication?
-
In a book entitled Cheaters Always Prosper: 50 Ways to Beat the System without Being Caught, James Brazil (a pen name), a college student from the University of California, Santa Barbara, has...
-
You deposit $10,000 in a savings account that earns 7.5% simple interest per year. What is the minimum number of years you must wait to double your balance? Suppose instead that you deposit the...
-
Find the area enclosed by the line y = x 1 and the parabola y 2 = 2x + 6.
-
How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper reagent? (b) (a)
-
Write the mechanism of the reaction between p-hydroxyaniline and acetic anhydride to prepare acetaminophen.
-
What product would you expect from reaction of 1 equivalent of methanol with a cyclic anhydride, such as phthalic anhydride (1, 2-benzenedicarboxylic anhydride)? What is the fate of the second ?half?...
-
Over a period of four and half years an investment grows to $ 2 0 , 0 0 0 . ( a ) If money in this investment accummlated at a simple interest rate of 8 % , what was the initial amount for the...
-
It is December 3 1 , 2 0 2 3 . Lincoln has an asset ( Basis $ 1 0 , 0 0 0 ; FMV = $ 4 0 , 0 0 0 ) . The gainon the asset is subject to depreciation recapture and will result in $ 3 0 , 0 0 0 in...
-
Define and explain the four basic functions that constitute the management process? What is a partnership? List four advantages and disadvantages of operating a business as partnership? Give an...

Study smarter with the SolutionInn App


