Question: Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the nitrogen and a negative charge on the oxygen.
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the nitrogen and a negative charge on the oxygen. Explain what effect you expect this to have on the melting point of alanine.
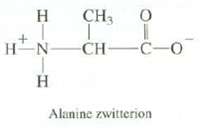
CH3 0 T HN CH- HINIH I -C-0 Alanine zwitterion
Step by Step Solution
★★★★★
3.44 Rating (173 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The zwitterions form of al... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
15-C-O-O-C (44).docx
120 KBs Word File


