Question: Glucose is a typical carbohydrate. What functional groups are present in glucose? What would you predict about the water solubility of glucose, oiler a reason
Glucose is a typical carbohydrate. What functional groups are present in glucose? What would you predict about the water solubility of glucose, oiler a reason why nature uses carbohydrates rather than alkenes as an energy source?
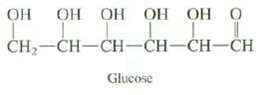
-- CH-CH-CH-CH-CH-CH Glucose
Step by Step Solution
3.41 Rating (170 Votes )
There are 3 Steps involved in it
Glucose contains alcohol and aldehyde functional groups Because it contai... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-O-C (43).docx
120 KBs Word File


