Question: Convert the following line-bond structures into molecularformulas: CH CH- (a) (b) . Hi H. c=C Vitamin C Aspirin (acetylsalicylic acid) (ascorbic acid) (c) H. -
Convert the following line-bond structures into molecularformulas:
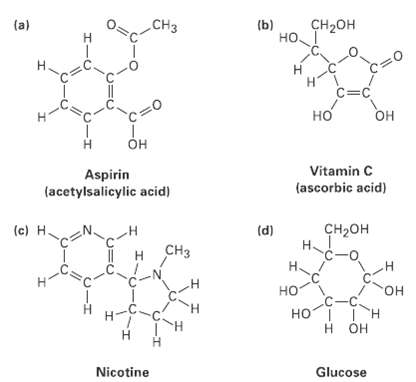
CH CH- (a) (b) . Hi H. c=C Vitamin C Aspirin (acetylsalicylic acid) (ascorbic acid) (c) H. - (d) H~ CH c-o H. . HOI I H H OH Nicotine Glucose
Step by Step Solution
3.39 Rating (165 Votes )
There are 3 Steps involved in it
In molecular formulas of organic molecules carb... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-C (228).docx
120 KBs Word File


