Electrostatic potential maps of (a) Formaldehyde (CH2O) and (b) Methanethiol (CH3SH) is shown. Is the formaldehyde carbon
Question:
Electrostatic potential maps of
(a) Formaldehyde (CH2O) and
(b) Methanethiol (CH3SH) is shown. Is the formaldehyde carbon atom likely to be electrophilic or nucleophiic? What about the Methanethiol sulfur atom?Explain.
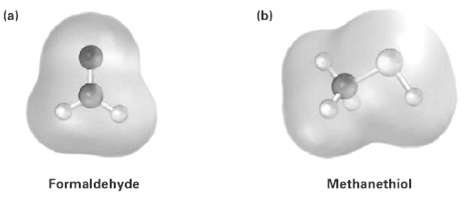
Transcribed Image Text:
(a) (b) Formaldehyde Methanethiol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
a The electrostatic potential map shows that the formaldehyde o...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid, p-methoxybenzoic acid or p-(methylthio) benzoic acid?Explain. Anisole (CGH5OCH3)...
-
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions?...
-
The molecular electrostatic potential maps for LiH and HF are shown here. Does the apparent size of the hydrogen atom (shown as a white sphere) tell you whether it is an electron acceptor or an...
-
Many areas have attempted to increase the amount of recycled waste lubricating oil by requiring service stations to serve as collection centers or by instituting deposit-refund systems. On what...
-
How can asset allocation help you build an investment program to reach your financial goals?
-
The rigid beam is supported by the three suspender bars. Bars AB and EF are made of aluminum and bar CD is made of steel. If each bar has a cross-sectional area of 450 mm 2 , determine the maximum...
-
Consider the gasoline mileage performance data in Table B.3. a. Use the all-possible-regressions approach to find an appropriate regression model. b. Use stepwise regression to specify a subset...
-
The following table shows the annual returns (in percent) and summary measures for the Vanguard Energy Fund and the Vanguard Health Care Fund from 2005 through 2009. a. Which fund had the higher...
-
Translate the following code into MIPS code. int Test (int i, int j) { int a; a = i + j + 5; a = Leaf (a + 1) + Leaf (a); return a; } int Leaf (int b) { int c; c = b+b; return g; } Assume variables a...
-
As part of a major plant renovation project, the industrial engineering department has been asked to balance a revised assembly operation to achieve an output of 240 units per eight- hour day. Task...
-
The following structure represents the carbocation intermediate formed in the addition reaction of HBr to two different alkenes. Draw the structures ofboth.
-
Look at the following energy diagram: (a) Is ?G? for the reaction positive or negative? Label it on the diagram. (b) How many steps are involved in the reaction? (c) How many transition states are...
-
On May 1, 2022, Lubins Heavy Equipment sold a piece of equipment to Perry Products, Inc., at a selling price of $4,850,000. Lubins agreed to accept a 10-month, 8% note with interest due on its...
-
Edgar was a computer programmer at Fastco; he has muscle spasms that prevent him from walking and is limited in access to areas where he can use his wheelchair. Edgar applied for a job at a...
-
1.At the beginning of Caste , author Isabel Wilkerson compares American racial hierarchy to a dormant Siberian virus. What are the strengths of this metaphor? How does this comparison help combat the...
-
Please be sure to view all of the required videos and respond to questions provided in the description of the assignment dropbox. You should compose your answer using Word (or another word processing...
-
Organic Green Pastures, a producer of chemicals for farming, whose headquarters (HQ) is in the United States, is considering setting a subsidiary in Malaysia. For the first year, Organic Green...
-
Raj works at an upscale department store. Because of his religion, Raj regularly wears a headscarf. Several customers and employees complain to management that they feel uncomfortable around Raj...
-
Your best friend is holding a black and a white marble, and asks you to open your hands and close your eyes. (a) He deposits one marble in each of your hands, and asks you to close your hands before...
-
Explain why each of the following is either a private good or a public good: traffic lights, in line skates, a city park, a chicken salad sandwich, a tennis racket, national defense, a coastal...
-
Delocalized molecular orbitals are found in (a) H 2 ; (b) HS - ; (c) CH 4 ; (d) CO 3 2- .
-
Provide a detailed mechanism for each of the following reactions. Include contributing resonance structures and the resonance hybrid for the arenium ion intermediates. (a) (b) (c) HNO HSO NO2 Br Bra,...
-
Provide a detailed mechanism for the following reaction. H,SO + H2O
-
One ring of phenyl benzoate undergoes electrophilic aromatic substitution much more readily than the other. (a) Which one is it? (b) Explain your answer.
-
Question: d. ?During December, Ingrid Legal Services provided legal services, and the client prepaid $7,000. ?Ingrid Legal Services recorded this amount as Unearned Revenue. The job will take several...
-
Question: A company had the following purchases and sales during the month of November:\table[[Date,Activities,Units Acquired at Cost,Units Sold at Retail],[November 1,Beginning inventory,5 units @...
-
Question: Tania, Inc. issued a note payable with principal and interest due at the end of the term of the note. Details about the note payable are as follows:Issuance date: October 1, fear...

Study smarter with the SolutionInn App


