Question: Knowing that at the instant shown bar DE has a constant angular velocity of 18 rad/s clockwise, determine (a) The acceleration of point B, (b)
Knowing that at the instant shown bar DE has a constant angular velocity of 18 rad/s clockwise, determine
(a) The acceleration of point B,
(b) The acceleration of point G.
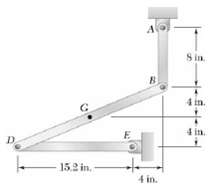
-15.2 in.- A B 4 in. 8 in. 4 in.
Step by Step Solution
3.40 Rating (175 Votes )
There are 3 Steps involved in it
19 A S E B 10 Velocity analysis DE 18 rads VD DE DE 15218 2736 in... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
4-E-M-E-VM (2151).docx
120 KBs Word File


