Examine the reaction network and the reaction kinetics for ethylene oxide production in Appendix B, Section B.6.2.
Question:
Examine the reaction network and the reaction kinetics for ethylene oxide production in Appendix B, Section B.6.2. What conditions will maximize the selectivity for ethylene oxide? Sketch concentration profiles for the case when this reaction is run in a packed bed modeled as a PFR.
Data from section B.6.2

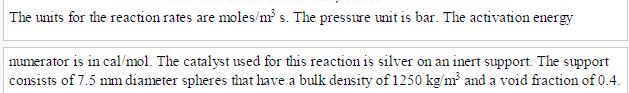
Transcribed Image Text:
The pertinent reactions (adapted from Stoukides and Pavlou [3]) are as follows: CH, +0,5 0, CHO + CH, 30, 2CO, + 2H,O CHO+2.5 02 2CO + 2HO The kinetic expressions are, respectively. 1 1.96 exp(-2400/RTP 1 +0.00098 exp(11,200/RT)Petkykne 0.0936 exp(-6400/RT)Peme Petinylene 12 1+0.00098 exp(11,200/RT)pet 0.42768 exp(-6200/RT)Phone ide 13 1+0.000033 exp(21,200/RT)Perrie (B.6.1) (B.6.2) (B.6.3) (B.6.4) (B.6.5) (B.6.6) The units for the reaction rates are moles/m s. The pressure unit is bar. The activation energy
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Analysis Synthesis And Design Of Chemical Processes
ISBN: 9780132618120
4th Edition
Authors: Richard Turton, Richard C. Bailie, Wallace B. Whiting, Joseph A. Shaeiwitz, Debangsu Bhattacharyya
Question Posted:
Students also viewed these Engineering questions
-
Examine the reaction network and the reaction kinetics for drying oil production in Appendix B, Section B.4.2. What conditions will maximize the selectivity for drying oil? Sketch concentration...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
The dimension of a services cape where the background characteristics of the environment that are meant to satisfy our five senses and include things such as temperature, lighting, noise, music, and...
-
The variance summary for Bell Computers is as follows: Required Prepare a journal entry to close the variance accounts at Bell Computers. Assume that the total variance is not material and is closed...
-
Does scheduling according to earliest due date tend to minimize average flowtime or average tardiness?
-
The following table shows data from a dehumidification process using aqueous solution of lithium chloride ( \(\mathrm{LiCl}\) ) in a packed tower. The desiccant leaving the dehumidifier is passed...
-
Kopke Company, organized in 2012, has these transactions related to intangible assets in that year: Jan. 2 Purchased a patent (5-year life) $280,000. Apr. 1 Goodwill acquired as a result of purchased...
-
a) A syrup is a mixture of sugar and water. Syrup A and B are to be mixed together. Syrups A and B contain 3.5 wt% and 4.2 wt% sugar, respectively. The resultant mixture is added with 17.5 kg of pure...
-
Investigate the effect of increasing reactor volume on toluene conversion. Plot the results. Problems 23. 12-23.16 investigate the performance of the reactor section for a noncatalytic process for...
-
For the situation in Example 23. 3, if all of the reactions take place, what conditions maximize the selectivity for \(P\)? Example 23.3 Consider the reaction scheme given in Equation (23.1) where P...
-
Show that if { (, (1, . . . , (n} is an orthogonal set of functions on [a, b] with respect to the weight function w, then {(0, (1, . . . , (n} is a linearly independent set.
-
. Consider sequences as given below, find y[n] = x[n] h [n] using formula-based method. (3 Marks x[n] = {-2 h[n] = (5 -4 -2}, 3}, -1 n 1 0 n1
-
8. Ahmed is working in a leading bank in Oman, but he decided to leave the organization after case to Mohammed, his boss but he didn't take any corrective action. However, Ahmed Is a well-known...
-
c) Start with the Glosten-Milgrom Model with the following parameters: 1 V = 60, V = 90, =- 3 = where V is the value of the stock, u is the proportion of informed traders in the market, and is the...
-
Four chairs (A, B, C, and D) are placed in a circle: A next to B. B next to C, C next to D, and D next to A. Each chair may be occupied (1) or empty (0). Express the outputs in a Truth Table and give...
-
Stackit Ltd. manufactures three sizes of wooden bookcases. The information relating to the production of the three bookcases for next year is as follows: Selling Price Cost of wood Direct labour...
-
You have been offered the opportunity to purchase a franchise of Sunshine Juice Stores. You will have to pay $258,635 for the initial investment in the store and its equipment, plus $30,000 per year...
-
What are the before image (BFIM) and after image (AFIM) of a data item? What is the difference between in-place updating and shadowing, with respect to their handling of BFIM and AFIM?
-
What term describes the method by which a fixed capital investment can be used to reduce the tax that a company pays?
-
Discuss the differences and similarities between interest, inflation, and the time value of money.
-
The value of the Chemical Engineering Plant Cost Index (CEPCI) at the beginning of 2016 is 557. If at the same time next year the value has risen to 594, what will be the average inflation rate for...
-
Shifted to MacOS from Windows, i was using pyodbc to fetch, insert, update data from MS access. however, I just realise it is not working in MacOS any suggestion for me to interact with database...
-
The information contained in the table below shows the expected return and standard deviation for the market and Treasury Bills. Market Data Treasury Bills S&P 500 Rate of Return Standard Deviation...
-
Assuming that the March 1 work in process inventory includes $18,000 of direct materials, determine the increase or decrease in the cost per equivalent unit for direct materials and conversion...

Study smarter with the SolutionInn App


