If the wall in Fig. 4.29 is 4 m long, calculate the total force on the wall
Question:
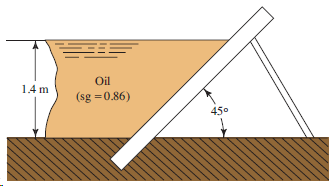
Transcribed Image Text:
Oil 14 m (sg =0.86) 450
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
F R 468 kN h p 23 h ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
If the cord attached to the vertical wall in Fig. 8.44 were horizontal (instead of at a angle), what would the tensions in the cords be? cord 1 45 30cord 2 1m 1.5 kg
-
Calculate the total force on the bottom of the closed tank shown in Fig. 4.23 if the air pressure is 52 kPa(gage). Air 0,50 m Oil (sg -0.85) 0.75 m Water 18 m 1.2 m
-
In Fig. E6.7 assume that there is no friction force on the 20.0-N block that sits on the tabletop. The pulley is light and frictionless. (a) Calculate the tension T in the light string that connects...
-
Builder Products, Incorporated, uses the weighted-average method in its process costing system. It manufactures a caulking compound that goes through three processing stages prior to completion....
-
Ryou Kato owns Kato Blankets. Ryou asks you to explain how he should treat the following reconciling items when reconciling the company's bank account. 1. Outstanding checks. 2. A deposit in transit....
-
Writing large letters. A large letter H can be produced like this: It can be declared as a string literal like this: final string LETTER_H = "* *%n* *%n*****%n* *%n* *%n"; Print the string with...
-
Develop the questionnaire in Internet Exercise 8-21 using an electronic questionnaire design package that is available for free (see the websites mentioned in this chapter in the subsection entitled...
-
The cash account of Dixon Products reveals the following information: The bank statement for May contains the following information: A comparison of detailed company records with the bank statement...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...
-
Reduction of evaporation losses by transpiration (Fig. 11B.7), it is proposed to reduce the rate of evaporation of liquefied oxygen in small containers by taking advantage of transpiration. To do...
-
The wall shown in Fig. 4.28 is 20 ft long. (a) Calculate the total force on the wall due to water pressure and locate the center of pressure; (b) Calculate the moment due to this force at the base of...
-
Explain the difference between optimistic and pessimistic locking.
-
Assume Coffee House, Inc., opened an office in Cocoa Beach, Florida. Further assume that Coffee House incurred the following costs in acquiring land, making land improvements, and constructing and...
-
Exhibit A.2 lists Fayols 14 principles of management, first published in 1916. Are they as useful today as they were then? Why or why not? When are they most, and least, useful? EXHIBIT A.2 Fayol's...
-
What are the welding defects? Explain the causes and remedies.
-
Explain briefly about soldering.
-
Enumerate and explain various allowances provided for pattern making.
-
Amigos Burrito Inc. sells franchises to independent operators throughout the northwestern part of Brazil. The contract with the franchisee includes the following provisions. 1. The franchisee is...
-
What things might managers in an organization do that could unintentionally discourage innovation by their employees? How can innovation be encouraged?
-
Swifty company is a publicly held corporation whose $1 par value stock is actively traded at $30 per share. The company issued 3400 shares of stock to acquire land recently advertised at $93000. When...
-
From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data Tables), calculate the standard enthalpy of formation of H 2 S(g) and of FeS 2 (s): AR(kJ mol) Fe(s) + 2H2S(g) ...
-
Which of Ne or Ar has the larger van der Waals parameter b? Explain your reasoning.
-
You have calculated the pressure exerted by ethane using the ideal gas law and the RedlichKwong equations of state. How do you decide if the repulsive or attractive part of the molecular potential...
-
A 1 kg metal block is heated to 200 C and then dropped into a thermally isolated container with 4 kg of water and 100 gr of ice both at a temperature of 0 C. If the specific heat of water is cw=4186...
-
Why are both ER Diagrams and Relational Modeling needed when a database system is developed?
-
Which data model provides a user with the best "program-data independence"? A. Hierarchial Model B. Network Model C. Relational Model D. Entity Relationship Model Why?

Study smarter with the SolutionInn App


