Consider atoms with the following electron configurations: Which atom has the largest first ionization energy, and which
Question:
Consider atoms with the following electron configurations:
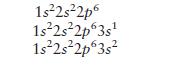
Which atom has the largest first ionization energy, and which has the smallest second ionization energy? Explain your choices.
Transcribed Image Text:
1s2s2p6 1s2s2p63s 1s2s2p 3s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The atom with the largest value of I is the one with the configu ration 1s2...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Graph f(x) - x, g(x) x +3 and (x) x5. Calculate the derivatives of f, g and h.
-
Identify the outer electron configurations for the (a) alkali metals, (b) alkaline earth metals, (c) halogens, (d) noble gases. For each electronic configuration given, choose the electronic...
-
Consider the formation of atomic hydrogen in the reaction c + H+ = H, where e is an electron, as the adsorption of an electron on a proton H+. (a) Show that the equilibrium concentrations of the...
-
Consider a situation with J identical firms that have marginal abatement cost functions for j=1,,J. The marginal damage function is equal to D'(E)=d.EDetermine the optimal allocation and the optimal...
-
Where g is the gravitational field strength, determine the value of T. T = 27V V/gA
-
Assume that the HOH bond angles in various crystalline hydrates have been measured to be 108 , 109 , 110 , 103 , 111 , and 107 . Give your estimate of the correct bond angle and its 95%...
-
Using only the factor formulas given in Table 2.6, derive Equation 7.6 starting with Equation 7.3. TABLE 2.6 Summary of Discrete Compounding Interest Factors. To Find Given Factor Symbol Name P F...
-
Comprehensive Problem; Second Production DepartmentWeighted-Average Method Old Country Links Inc. produces sausages in three production departmentsMixing, Casing and Curing, and Packaging. In the...
-
An economic consultant for a fast-food chain has been given a random sample of the chains service workers. In her model, A, the length of time y between the time the worker is hired and the time a...
-
Predict the trend in radius of the following ions: Be 2+ , Mg 2+ , Ca 2+ , and Sr 2+ .
-
The first ionization energy for phosphorus is 1060 kJ/mol, and that for sulfur is 1005 kJ/mol. Why?
-
Mary Lou Golini appeals the circuit courts affirmance of a probate court order admitting a copy of Willie Mae Arants last will to formal probate and appointing Melvin R. Bolton personal...
-
Jake has $44,000 invested in Vanguard Money Market Mutual Fund for 71 days at a 7-day yield of 3.62%. How much interest will he receive in 71 days?
-
5. A 460 N force pulls due north on a crate. What other force must act on the crate if: a) the net force on the crate is 386 N due south? b) the net force on the crate is 152 N due north? a) 3 marks
-
Conservation of Energy Applied to a Swimming Pool You are asked to design a heating system for a variety of rectangular swimming pools that are 2 m deep, 10 m wide, and the length can vary from 10 m...
-
The ball of a pinball machine is accelerated from rest to 8 in/s in a distance of 7.7 in. Determine the acceleration of the ball.
-
How many red photons of wavelength 806 nm would it take to raise the temperature of 1g of water 1 degree Celsius? How many blue photons of 428 nm would it take to do the same?
-
Assume that General Dynamics, which manufactures high-technology instruments for spacecraft, is considering the sale of a navigational unit to a government agency in India that wishes to launch its...
-
Complete the equations for the following equilibria and calculate Keq where the Keq expression includes [HO]. Be sure to enter Keq in proper scientific notation. (a) ammonia (acting as a base) reacts...
-
For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or Appendix 2B, with the exception of the reduction and oxidation of water at pH = 7: The anode of an...
-
(a) When the pH of 0.10 m HClO 2 (aq) was measured, it was found to be 1.2. What are the values of K a and pK a of chlorous acid? (b) The pH of a 0.10 m propylamine, C 3 H 7 NH 2 , aqueous solution...
-
Balance each of the following skeletal equations by using oxidation and reduction half-reactions. All the reactions take place in acidic solution. Identify the oxidizing agent and reducing agent in...
-
The goal of this project is to design a suitable and sustainable cooling system at Tambient=40C for a 400W ASIC and its 4qty x 5W memory in a 1U (1.75in Tall) system using water (Tinlet=40C) and air...
-
1. Determine the toggle positions and the minimum transmission angle. Link 2 A2 02. Link 3 A B Link 1 Link 3 B1 Link 4 04
-
2. The graphs show the performance of a crystalline silicon solar panel. Figure 1 Current (A) 14 12 20% 642 10 8 0 Figure 2 600 16 550 14 500 12 450 400 350 300 250 200 150 Power (W) Current (A) 10 8...

Study smarter with the SolutionInn App


