Predict the standard cell potential and calculate the standard reaction Gibbs free energy for galvanic cells having
Question:
Predict the standard cell potential and calculate the standard reaction Gibbs free energy for galvanic cells having the following cell reactions: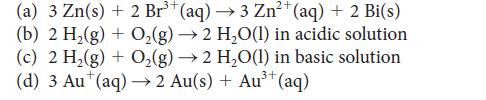
Transcribed Image Text:
(a) 3 Zn(s) + 2 Br³ (aq) →3 Zn²+ (aq) + 2 Bi(s) (b) 2 H₂(g) + O₂(g) → 2 H₂O(l) in acidic solution - (c) 2 H₂(g) + O₂(g) → 2 H₂O(1) in basic solution (d) 3 Au* (aq) → 2 Au(s) + Au³+ (aq) 3+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Here are the predicted standard cell potentials and calculated standard reaction Gibbs free energies ...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The standard reduction potentials of the following half-reactions are given in Appendix E: (a) Determine which combination of these half-cell reactions leads to the cell reaction with the largest...
-
Suppose that you are in an engineering competition in which commercial power sources are prohibited. You might decide to use a Daniell cell to power a model electric car. You will need to know the...
-
Fill in the blanks in the table below. The problem is a "puzzle" so the blanks are not necessarily filled in sequentially. Hint: Determine the total fixed cost first. Instructions: Round your answers...
-
You are looking at buying a piece of real estate and you intend to borrow as much as you possibly can from a bank to buy the property. The bank you are dealing with has a requirement that the LVR for...
-
Standard-costing with beginning and ending work in process. Paquitas Pearls Company (PPC) is a manufacturer of knock off jewelry. Paquita attends Fashion Week in New York City every September and...
-
Six Jobs are to be processed through a two-step operation. The first operation involves sanding, and the second involves painting. Processing times are as follows: Determine a sequence that will...
-
Identify three selling situations where a memorized sales presentation may be appropriate. Explain why they would be appropriate.
-
1. What are the ethical issues in this case? 2. What are some reasons the decision maker in this case might be inclined to go along? Not go along? 3. If you were the decision maker, how would you...
-
How does the Agile methodology accommodate evolving requirements and stakeholder feedback throughout the software development lifecycle, thereby enabling teams to embrace change as a competitive...
-
Dinitrogen monoxide, N 2 O, reacts with water to form hyponitrous acid, H 2 N 2 O 2 (aq), in a Lewis acidbase reaction. (a) Write the chemical equation for the reaction. (b) Draw the Lewis structures...
-
Calculate the pH and pOH of each of the following aqueous solutions of strong acid or base: (a) 0.0356 m HI(aq); (b) 0.0725 m HCl(aq); (c) 3.46 * 10 3 m Ba(OH) 2 (aq); (d) 10.9 mg of KOH dissolved...
-
A particle is launched with a horizontal velocity v 0 = 0.55 m/s from the 30 position shown and then slides without friction along the funnel-like surface. Determine the angle which its velocity...
-
On January 1, 2020 - A government passes a budget for the General fund with the following: Estimated Property tax $18,000,000; estimated grant revenue of 1,000,000. The GENERAL FUND estimates it will...
-
Your preparer tax identification number (PTIN) is PO0389571. Your Firm ik For) Jones and Miller's address is 230 Hollister Avenue - San Francisco, CA 94131 and its phone number is (650) 686-7777....
-
Tasks For each of the following tasks you are to complete the following steps provided to demonstrate your ability to perform each of the following financial calculations: 1. Goods and services tax...
-
2-stage DDM model: g = 0.2459 for 3 years; g =Rf = 2% afterward Intrinsic value = D/(1+R) + D(1+R) + (D3+P3)/(1+R) = 0.2071*1.2459/1.0528+ 0.2071*1.24592/(1.0528)2 + (0.2071* 1.24593+ 0.2071*...
-
Account Titles cash in bank ordinary share capital share premium-ordinary preference share share premium-ps ppe-building ppe-land mortgage payable ppe-office furnitures & Notes Payable Smallspoon...
-
What might the components of a balanced scorecard be for a municipality? For a not-for-profit hospital?
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
When the following compound is treated with excess methyl iodide, a quaternary ammonium salt is obtained that bears only one positive charge. Draw the structure of the quaternary ammonium salt. *NH2
-
Draw a mechanism for the following transformation: 'CI Z Z
-
In Problem 22.38, we saw an intramolecular example of a malonic ester synthesis using excess base and 1,4-dibromobutane. If this dibromide is used in an acetoacetic ester synthesis, an intramolecular...
-
Discuss the general properties of coverage offered under a comprehensive personal liability insurance policy. In your answer, address the exposures this type of coverage addresses and the nature of...
-
Given the six years of percentage linear return from Stock 1, 2 and 3 in the following table Year Stock 1 return Stock 2 return Stock 3 return 2013 0.10 0.20 -0.05 2014 -0.15 -0.20 -0.3 2015 0.20...
-
6. Justin wants to purchase a $250,000 5-year term life insurance policy. He is 24 years old. What is his annual premium? 7. Eight years ago, Michael purchased a $150,000, 20-year payment life...

Study smarter with the SolutionInn App


