Question: Use data from Appendix 2B to decide whether chlorine gas will oxidize Mn 2+ to form the permanganate ion in an acidic solution. 2B STANDARD
Use data from Appendix 2B to decide whether chlorine gas will oxidize Mn2+ to form the permanganate ion in an acidic solution.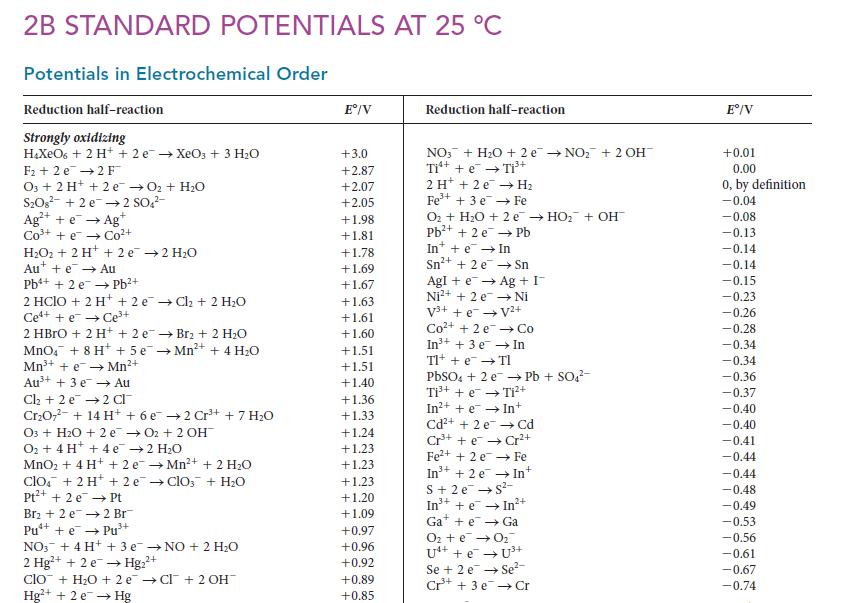
2B STANDARD POTENTIALS AT 25 C Potentials in Electrochemical Order Reduction half-reaction Strongly oxidizing HXeO6 + 2 H+ + 2 e XeO3 + 3 HO F+2 e 2 F O3 + 2 H+ + 2e O + HO SO8 +2e 2 SO4- Ag+ + e Ag C t HO + 2H+ + 2e 2 HO Aue Au Pb+ + 2 e Pb+ 2 HClO + 2 H+ + 2 eCl + 2 HO Ce++eCe+ 2 HBrO + 2H+ + 2 e Br2 + 2 HO MnO4 + 8 H+ + 5e Mn+ + 4HO Mn+ + eMn+ Au+ + 3 e Au Cl + 2 e 2 Cl CrO7- + 14 H+ + 6 e cot O3 + HO + 2 e O + 4H+ + 4e 2 HO MnO + 4H+ + 2e Mn+ + 2 HO clo + 2 H+ + 2 eClO3 + HO Pt+ + 2e Pt Br + 2 e Pu+ + ePu+ 2 Br 3+ 2 Cr+ + 7 HO O2 + 2 OH NO3 + 4H+ + 3 eNO + 2 HO 2 Hg+ + 2e Hg+ 2+ clo + HO +2e Cl + 2 OH Hg+ + 2e Hg E/V +3.0 +2.87 +2.07 +2.05 +1.98 +1.81 +1.78 +1.69 +1.67 +1.63 +1.61 +1.60 +1.51 +1.51 +1.40 +1.36 +1.33 +1.24 +1.23 +1.23 +1.23 +1.20 +1.09 +0.97 +0.96 +0.92 +0.89 +0.85 Reduction half-reaction NO3 + HO + 2e NO + 2 OH- Ti+ + e Ti+ 2 H + 2e H Fe+ + 3 e Fe O + HO + 2e HO + OH Pb+ + 2 e Pb In+ + e In Sn+ + 2 e Sn Ag + I Ni AgI + e Ni+ + 2 e V+ + e V+ Co+ + 2e In+ + 3 e Tl+ + e TI Co In PbSO4 + 2 ePb + SO4- Ti+ + e Ti+ In++eIn+ Cd+ + 2 e Cd Cr+ + eCr+ Fe+ + 2e Fe In +2 e S+ 2e S- + In +eIn2+ Gae Ga O + e 0 U4+ + e Se + 2 eSe- Cr+ + 3e Cr E/V +0.01 0.00 0, by definition -0.04 -0.08 -0.13 -0.14 -0.14 -0.15 -0.23 -0.26 -0.28 -0.34 -0.34 -0.36 -0.37 -0.40 -0.40 -0.41 -0.44 -0.44 -0.48 -0.49 -0.53 -0.56 -0.61 -0.67 -0.74
Step by Step Solution
3.40 Rating (159 Votes )
There are 3 Steps involved in it
2 Clg will n... View full answer

Get step-by-step solutions from verified subject matter experts


