Question: Use the data in Appendix 2A to calculate the standard reaction enthalpy for the reaction of magnesium carbonate with hydrochloric acid: MgCO3 (s) + 2
Use the data in Appendix 2A to calculate the standard reaction enthalpy for the reaction of magnesium carbonate with hydrochloric acid:![]()
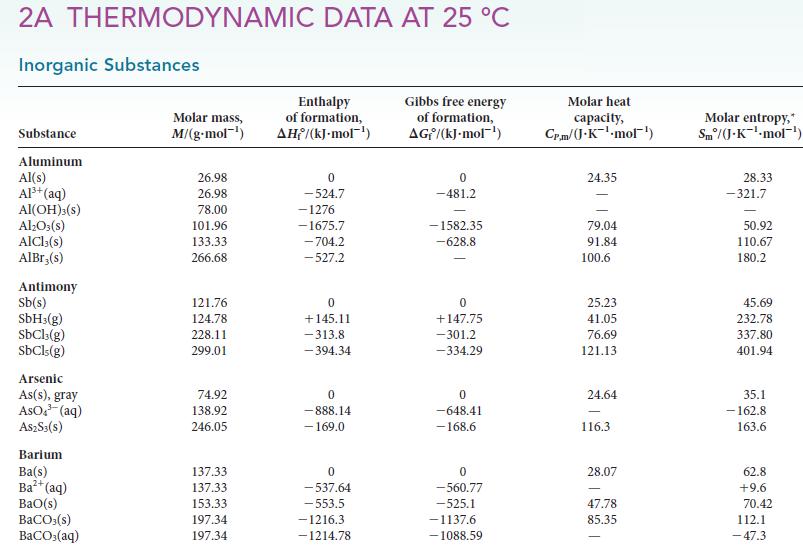
MgCO3 (s) + 2 HCI (aq) MgCl (aq) + HO(1) + CO(g)
Step by Step Solution
★★★★★
3.34 Rating (163 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

MgCO3s 2HClaq AH for MgCO3s MgClaq HO CO2g 10958 K... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


