Write the half-reactions, the balanced equation for the cell reaction, and the cell diagram for each of
Question:
Write the half-reactions, the balanced equation for the cell reaction, and the cell diagram for each of the following skeletal equations: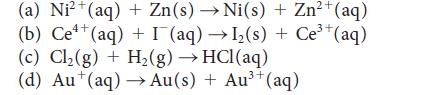
Transcribed Image Text:
(a) Ni²+(aq) + Zn (s) → Ni(s) + Zn²+(aq) Ce4+ (b) Ce¹¹ (aq) + I (aq) →1₂(s) + Ce³+(aq) (c) Cl₂(g) + H₂(g) → HCl(aq) (d) Au¹(aq) →→ Au(s) + Au³+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a anode Zns Zn aq 2e cathode Ni aq 2 eNis 2 2 overall Ni aq Zns Nis Zn aq 2 Z...View the full answer

Answered By

Fahad P
During my masters I realized teaching is the the key to understand physics deeper, that is why I started teaching as a part time work. I got chance to work at few coaching centers at my home town to teach physics to students, it was nice but I could understand that teaching students individually is more satisfying for me, because we can get their full attention and once they start loving our classes they will get involved in more and more and that experience we get during those sessions become unforgettable .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write the half reactions for the electrolysis of the elements listed in Exercise 3.
-
The nickelcadmium cell is rechargeable. The half equations for the electrode reactions are: Cd(OH) 2 + 2e Cd + 2OH E = 0.81 V NiO 2 + 2H 2 O + 2e Ni(OH) 2 + 2OH E = +0.49 V a. Which of these...
-
Write a balanced equation for each of the following reactions or reaction sequences. (a) The reaction catalyzed by PFK-2 (b) The conversion of 2 moles of oxaloacetate to glucose (c) The conversion of...
-
A machine was sold in December 20x3 for $13,000. It was purchased in January 20x1 for $19,000, and depreciation of $16,000 was recorded from the date of purchase through the date of disposal....
-
Customer profitability Ring Delights is a new company that manufactures custom jewelry. Ring Delights currently has six customers referenced by customer number 01, 02, 03, 04, 05 and 06. Besides the...
-
What processes are supported by information provided by IT asset management?
-
How can the strength of association be measured in a multiple regression model?
-
On December 31, 2012, Paxon Corporation acquired 90 percent of the outstanding common stock of Saxon Company for $1,620 million cash. The fair value of the 10 percent noncontrolling interest in Saxon...
-
Nora Incorporated sells a single product for $22. Variable costs include $8.58 for each unit plus a 8% sales commission. Fixed costs are $155,290 per month. Required: a. What is the contribution...
-
For each reaction that is spontaneous under standard conditions (that is, K > 1), write a cell diagram, determine the standard cell potential, and calculate G for the reaction: (a) 2 NO3 (aq) + 8 H...
-
Decide whether a precipitate will form when each of the following pairs of solutions are mixed: (a) 5.0 mL of 0.10 m K 2 CO 3 (aq) and 1.00 L of 0.010 m AgNO 3 (aq); (b) 3.3 mL of 1.0 m HCl(aq), 4.9...
-
Polaris financial statements and notes in Appendix A provide evidence of growth potential in its sales. Required 1. Identify at least two types of costs that will predictably increase as a percent of...
-
CASH FLOW EXERCISE Balance sheet, income statement and cash flow. The Statement of financial position of YYY for 2018 is the following: Assets Equity Non-current assets 160 Equipment 200 (Accumulated...
-
You may not buy a home or need a mortgage for several years, but it's important to understand some of what that entails. Complete this worksheet with your first home in mind. I hope to buy my home in...
-
1-Using number from 5/31/2021, compute cash collected from customers: Sales Revenue 32,376 Decrease in Accounts Receivable Cash Collected from Customers 2-Using the numbers from 2016, compute cash...
-
The following spreadsheet contains monthly returns for Cola Co. and Gas Co. for 2013. Using these data, estimate the average monthly return and the volatility for each stock. (Click on the following...
-
1. ( 14 Points ) You are working for a private equity firm and your team is assigned the task of determining the enterprise value of a target firm. This target firms past and current Income...
-
In process costing, the volume of production for a period is likely to be much greater than the volume in inventory at any given point in time. This feature means that the value of inventory is a...
-
Huntingdon Capital Corp. is a competitor of Plazacorp and First Capital Realty. Huntingdon reported the following selected information (in millions):...
-
At 25C, the equilibrium constant for the dissociation of acetic acid, K a , is 1.75 10 5 . Using the DebyeHckel limiting law, calculate the degree of dissociation in 0.150 m and 1.50 m solutions...
-
From the data in Table 10.3 (see Appendix B, Data Tables), calculate the activity of the electrolyte in 0.200 m solutions assuming complete dissociation of a. KCl b. Na 2 SO 4 c. MgCl 2
-
Estimate the degree of dissociation of a 0.200 m solution of nitrous acid (K a = 4.00 10 4 ) that is also 0.500 m in the strong electrolyte given in parts (a)(c). Use the data tables to obtain ,...
-
Edge Corporation issues 20,000 shares of $5 stated value no par stock in exchange for land with an advertised price of $125,000 when the stock is actively trading for $6 per share. Journalize the...
-
Write a recurrence describing the number of times the algorithm compares two members of array A, measured as a function of the array length n. You do not need to solve the recurrence relation...
-
(a) Explain what is wrong with the given code. Describe the fault precisely by proposing a modification to the code. (b) If possible, give a test case that does not execute the fault. If not, briefly...

Study smarter with the SolutionInn App


