Codes, standards, and regulations: Go to the www.osha.gov web site and look up the OSHA regulation CFR
Question:
Codes, standards, and regulations: Go to the www.osha.gov web site and look up the OSHA regulation CFR 1910.119: Process Safety Management of Highly Hazardous Chemicals. Use Appendix A to determine the threshold quantities for the following chemicals. If your plant site exceeds this threshold quantity, then this standard applies.
a. Ammonia, anhydrous
b. Chlorine
c. Hydrogen fluoride
d. Propylene oxide
Appendix A
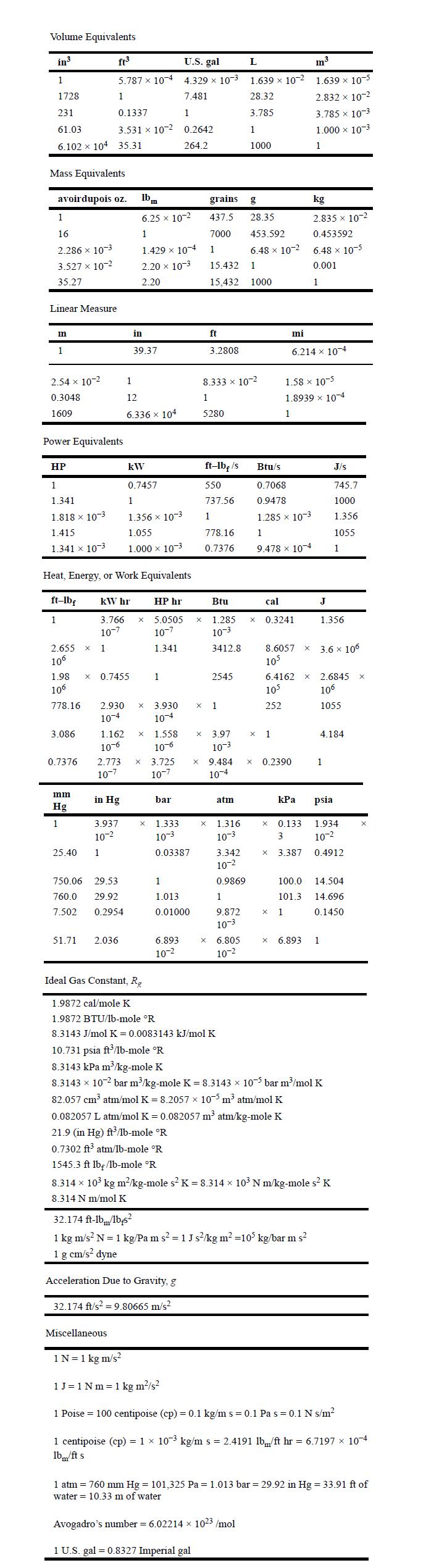
Transcribed Image Text:
Volume Equivalents in 1 1728 231 61.03 6.102 104 35.31 Mass Equivalents avoirdupois oz. 1 16 2.286 10-3 3.527 x 10-2 35.27 Linear Measure mn 1 2.54 x 10-2 0.3048 1609 1 Power Equivalents HP 1 1.341 1.818 10-3 1.415 1.341 x 10-3 2.655 X 1 10 ft U.S. gal 5.787 x 104 4.329 10-3 1 7.481 0.1337 1 3.531 x 10-2 0.2642 264.2 3.086 0.7376 Heat, Energy, or Work Equivalents ft-lbf kW hr mm Hg 1 1.98 X 0.7455 10 778.16 25.40 2.930 10-4 1.162 10-6 HP hr 3.766 X 5.0505 10-7 10-7 1.341 in Hg 3.937 10- 1 lbm 6.25 x 10-2 1 750.06 29.53 760.0 29.92 7.502 0.2954 1.429 x 10-4 2.20 10-3 2.20 51.71 2.036 in 39.37 1 12 6.336 x 104 kW 0.7457 1 1.356 x 10-3 1.055 1.000 10-3 X Miscellaneous X 2.773 X 3.725 10-7 10-7 1 X 3.930 10-4 1.558 10-6 bar 1.333 10-3 0.03387 1 1.013 0.01000 6.893 10-2 X grains 437.5 7000 1 15.432 15,432 X Acceleration Due to Gravity, g 32.174 ft/s = 9.80665 m/s ft 3.2808 ft-lb/s 550 737.56 1 778.16 0.7376 8.333 x 10-2 1 5280 2545 X 1 X 3.97 10-3 Btu 1.285 X 10-3 3412.8 9.484 10-4 Ideal Gas Constant, R 1.9872 cal/mole K 1.9872 BTU/lb-mole R 8.3143 J/mol K = 0.0083143 kJ/mol K atm X 1.316 10-3 3.342 10- 0.9869 1 9.872 10-3 X 6.805 10-2 L 1.639 x 10-2 28.32 3.785 1 1000 g 28.35 453.592 6.48 x 10-2 1 1000 X Avogadro's number = 6.02214 x 1023 /mol 1 U.S. gal = 0.8327 Imperial gal cal Btu/s 0.7068 0.9478 1.285 x 10-3 1 9.478 x 10-4 0.3241 x 1 8.6057 X 105 6.4162 105 252 0.2390 X mi 6.214 x 10-4 1.58 x 10-5 1.8939 x 10-4 1 kPa 0.133 3 X 3.387 X 1 0.082057 L atm/mol K = 0.082057 m atm/kg-mole K 21.9 (in Hg) ft/lb-mole R 0.7302 ft atm/lb-mole R 1545.3 ft lb/1b-mole R m 1.639 10-5 2.832 x 10-2 3.785 x 10-3 1.000 10-3 1 kg 2.835 x 10-2 0.453592 6.48 x 10-5 0.001 1 32.174 ft-lb/lbs 1 kg m/s N = 1 kg/Pa m s = 1 J s/kg m =10 kg/bar m s 1 g cm/s dyne J 1.356 X 2.6845 X 106 1055 3.6 x 106 X 6.893 1 1 10.731 psia ft/1b-mole R 8.3143 kPa m/kg-mole K 8.3143 10-2 bar m/kg-mole K = 8.3143 105 bar m/mol K 82.057 cm atm/mol K = 8.2057 105 m atm/mol K J/s 745.7 1000 1.356 1055 1 4.184 psia 100.0 14.504 101.3 14.696 0.1450 8.314 x 10 kg m/kg-mole s K = 8.314 x 10 N m/kg-mole s K 8.314 N m/mol K 1.934 10- 0.4912 1 N = 1 kg m/s 1 J = 1 Nm = 1 kg m/s 1 Poise = 100 centipoise (cp) = 0.1 kg/m s = 0.1 Pa s = 0.1 N s/m 1 centipoise (cp) = 1 x 103 kg/m s = 2.4191 lb/ft hr = 6.7197 10-4 lbm/ft s 1 atm = 760 mm Hg = 101,325 Pa = 1.013 bar = 29.92 in Hg = 33.91 ft of water = 10.33 m of water
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
How important it is to educate ourselves environmental issues impacting the future of tourism i.e., climate change The COVID-19 pandemic has changed the tourism industry forever and will impact our...
-
The block and tackle shown are used to raise a 600-N load. Each of the 60-mm-diameter pulleys rotates on a 10-mm-diameter axle. Knowing that the coefficient of kinetic friction is 0.20, determine the...
-
Use the following labor market data to determine the answers to (a) through (d): a. Is this tax progressive? Explain. b. What is the before-tax equilibrium wage rate? c. What effect does the tax have...
-
It looks to me that you have devoted everything to this company and they have treated you badly. Is that right?
-
The Carlson Department Store suffered heavy damage when a hurricane struck on August 31, 2003. The store was closed for four months (September 2003 through December 2003), and Carlson is now involved...
-
Three children must make fair division of a painting and sculpture left them by their mother. Using the Knaster Inheritance Procedure, the value each child places on the objects is shown below....
-
Safeguards: Classify the following safeguards as either preventive or mitigative. a. A safety instrumented system to shut down a process if an unsafe operating condition occurs. b. A foam system to...
-
Use the risk matrix in Table 1-14 to determine the risk level for the Bhopal incident. Estimate the severity category, the safety severity level, the likelihood, and the risk level. Table 1-14 Risk...
-
Use the super node technique to find Io in the circuit infigure. 12/6v -j10 ww j20 20
-
What is a Limited Liability Company, and how does it differ from a S-Corporation? Explain
-
The preferred stock of Dolphin Pools pays an annual dividend of $5.25 a share and sells for $48a share. The tax rate is 35 percent. What is the firm's cost of preferred stock?
-
Waffle House Inc. has a beta of 1.2, the expected return on the market is 13, and the risk-free rate is 2.8. What is the expected return on this stock?
-
The oldest child in a family of four children is twice as old as the yougest. The two middle children are 13 and 15 years old. If the average age of the children is 15.25, how old is the youngest...
-
Precision Cuts has a target debt-equity ratio of .48. Its cost of equity is 16.4 percent, and its pretax cost of debt is 8.2 percent. If the tax rate is 34 percent, what is the company's WACC?
-
Joys Construction is in its fourth year of business. Joy performs long-term construction projects and accounts for them using the completed-contract method. Joy built an apartment building at a price...
-
In a certain school district, 3% of the faculty use none of their sick days in a school year. Find the probability that 5 faculty members selected at random used no sick days in a given year.
-
Set up a spreadsheet to reproduce Figure 4-15. Add error bars: Follow the procedure on page 90. Use s y for the and + error. Figure 4-15 3 10 m 0.6154 1.3462" style="" class="fr-fic fr-dib"> A B D F...
-
Excel LINEST function. Enter the following data in a spreadsheet and use LINEST to find slope, intercept, and standard deviations. Use Excel to draw a graph of the data and add a trendline. Draw...
-
Using the linear calibration curve in Figure 4-13, find the quantity of unknown protein that gives a measured absorbance of 0.264 when a blank has an absorbance of 0.095. Figure 4-13 0.40 Unknown...
-
What is the return on stockholders' equity for a firm with a net profit margin of 22.86 percent, sales of $3,484,800, an equity multiplier of 1.2, and total assets of $2,939,980?
-
On 30 April 2022, Snake Ltd transferred inventory costing $50,000 to Carpet Ltd for $80,000. Carpet Ltd treated this item as plant. The item was still on hand at 30 June 2022. Carpet Ltd applied a...
-
CEO of start-up with 110 employees signs an agreement that if the start-up is purchased and she is replaced, she will receive $1,000,000. The start-up is indeed acquired and she is released and paid...

Study smarter with the SolutionInn App


