Conduct a safety review for the design of the system described in Example 11-2. This reactor is
Question:
Conduct a safety review for the design of the system described in Example 11-2. This reactor is used to polymerize ethylene oxide to form polyols.
Example 11-2
Transcribed Image Text:
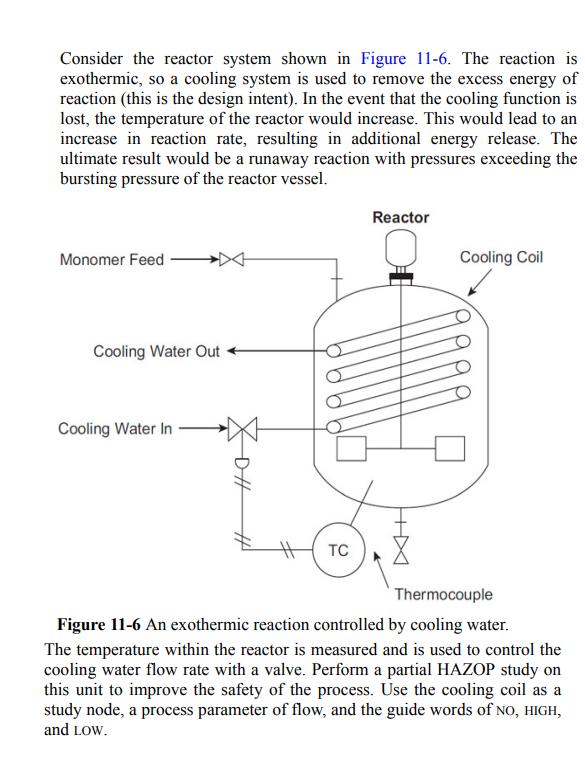
Consider the reactor system shown in Figure 11-6. The reaction is exothermic, so a cooling system is used to remove the excess energy of reaction (this is the design intent). In the event that the cooling function is lost, the temperature of the reactor would increase. This would lead to an increase in reaction rate, resulting in additional energy release. The ultimate result would be a runaway reaction with pressures exceeding the bursting pressure of the reactor vessel. Monomer Feed Cooling Water Out Cooling Water In TC Reactor X Cooling Coil Thermocouple Figure 11-6 An exothermic reaction controlled by cooling water. The temperature within the reactor is measured and is used to control the cooling water flow rate with a valve. Perform a partial HAZOP study on this unit to improve the safety of the process. Use the cooling coil as a study node, a process parameter of flow, and the guide words of NO, HIGH, and LOW.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
NO Cooling Water Flow Hazard If there is a complete failure in the cooling water flow NO flow the reactor temperature will continue to rise unchecked ...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
A good management practice is to set objectives before safety reviews are conducted. The objectives should include the timing for completing the objectives. Develop objectives for a safety review for...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Income statements for the current year and one year ago follow. Assume that all sales are on credit. For Year Ended December 31 Sales Cost of goods sold Other operating expenses Interest expense...
-
A thin steel wire of uniform cross section is bent into the shape shown, where arc BC is a quarter circle of radius R. Locate its center of gravity. 300 mm I80 am 100 m
-
The area between 1 and 2 for a standard normal distribution converted to a N(100, 15) distribution Ask you to convert an area from one normal distribution to an equivalent area for a different normal...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Earnhart Corporation has outstanding 3,000,000 shares of common stock of a par value of $10 each. The balance in its Retained Earnings account at January 1, 2012, was $24,000,000, and it then had...
-
One of your clients occasionally smokes cigarettes. They have told you their partner does not know and that they would be very mad with them if they found out. One day their partner asks you if they...
-
An operator needs to charge 5 kilograms of a catalyst into a batch reactor (Reactor A) 3 hours after the start of the batch. List 10 or more ways the operator can fail to perform this task correctly,...
-
The "fail safe" concept is used to specify the position (fail closed or fail open) of all process valves in the event of a utility failure. The specified fail open or fail closed puts the process in...
-
Mega Enterprises is in the process of negotiating an extension of its existing loan agreements with a major bank. The bank is particularly concerned with Mega's ability to generate sufficient cash...
-
2. Has Stitch Fix been able to generate and sustain a blue ocean in the online clothing retail industry (your strategy canvas should reflect your position on this question)? Remember, that a blue...
-
1. Office of Hawaiian Affairs (OHA's) unique internal and external political and legal issues, including the debate over Hawaiian Sovereignty 2. How Office of Hawaiian Affairs (OHA) related to other...
-
Jack has decided to terminate his employment with Gold's Manufacturing Company and would like to convert his group insurance policy to an individual policy. How long does he have? A. He must apply...
-
In regards to expectations, roles and responsibilities of team members, explain each of the following attributes of a well - rounded and valued team member. Adhere to policies and procedures...
-
As the lowest rates paid in the software, chemical, oil, and pharmaceutical industries are already well above minimum, any legislation to increase minimum wage would have little direct impact on...
-
Equipment acquired at the beginning of the year at a cost of $275,000 has an estimated residual value of $30,000 and an estimated useful life of 10 years. Determine (a) The depreciable cost, (b) The...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Why does the solubility of a salt of a basic anion increase with decreasing pH? Write chemical reactions for the minerals galena (PbS) and cerussite (PbCO 3 ) to explain how acid rain mobilizes...
-
Considering just acid-base chemistry, not ion pairing and not activity coefficients, find the pH and concentrations of species in 1.00 L of solution containing 0.020 mol arginine, 0.030 mol glutamic...
-
A solution containing 0.139 mmol of the triprotic acid tris(2-aminoethyl)amine 3HCl plus 0.115 mmol HCl in 40 mL of 0.10 M KCl was titrated with 0.490 5 M NaOH to measure acid dissociation...
-
A service-oriented business is struggling to manage its resources efficiently. How can ERP software assist in scheduling, allocating, and tracking resources such as personnel and equipment?
-
i. Mount Apo, the highest elevation in the Philippines, stands at 2,954 meters above sea level. Taal Volcano, the lowest elevation, is 311 meters above sea level. What is the difference between these...
-
How to find out NO geographic barriers in Online digital banking?

Study smarter with the SolutionInn App


