The irreversible endothermic vapor-phase reaction follows an elementary rate law CH3COCH3 CH2CO + CH4 A
Question:
The irreversible endothermic vapor-phase reaction follows an elementary rate law
CH3COCH3 → CH2CO + CH4
A → B + C and is carried out adiabatically in a 500-dm3 PFR. Species A is fed to the reactor at a rate of 10 mol/min and a pressure of 2 atm. An inert stream is also fed to the reactor at 2 atm, as shown in Figure P11-6B. The entrance temperature of both streams is 1100 K.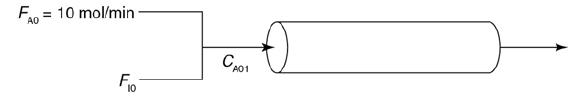
Additional information:
a. First derive an expression for CA01 as a function of CA0 and ΘI.
b. Sketch the conversion and temperature profiles for the case when no inerts are present. Using a dashed line, sketch the profiles when a moderate amount of inerts are added. Using a dotted line, sketch the profiles when a large amount of inerts are added. Qualitative sketches are fine. Describe the similarities and differences between the curves.
c. Sketch or plot and then analyze the exit conversion as a function of ΘI. Is there a ratio of the entering molar flow rates of inerts (I) to A (i.e., ΘI = FI0/FA0) at which the conversion is at a maximum? Explain why there “is” or “is not” a maximum.
d. What would change in parts (b) and (c) if reactions were exothermic and reversible with ΔHRx∘ = –80 kJ/mol and KC = 2 dm3/mol at 1100 K?
e. Sketch or plot FB for parts (c) and (d), and describe what you find.
Step by Step Answer:






