A laboratory analysis of aspirin determines the following mass percent composition: C 60.00%; H 4.48%; O 35.52%
Question:
A laboratory analysis of aspirin determines the following mass percent composition:
C 60.00%; H 4.48%; O 35.52%
Find the empirical formula.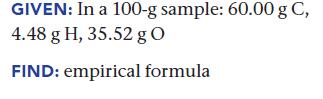
Transcribed Image Text:
GIVEN: In a 100-g sample: 60.00 g C, 4.48 g H, 35.52 g O FIND: empirical formula
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
6000 g x 448 g H x 1 mol C 1201 ge 1 mol H 10...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Estradiol is a female sexual hormone that is responsible for the maturation and maintenance of the female reproductive system. Elemental analysis of estradiol gives the following mass percent...
-
Fructose is a common sugar found in fruit. Elemental analysis of fructose gives the following mass percent composition: C 40.00%, H 6.72%, O 53.28%. The molar mass of fructose is 180.16 g/mol. Find...
-
Consider a beam with two perpendicular segments subjected to a concentrated load as shown in Fig. 9.52. Using two beam elements, determine the deflection and stresses in the two segments of the beam....
-
A piece of newly purchased industrial equipment costs $860,000 and is classified as seven-year property under MACRS. Calculate the annual depreciation allowances and end of the year book values for...
-
Springer Products wishes to borrow $80,000 from a local bank using its accounts receivable to secure the loan. The bank's policy is to accept as collateral any accounts that are normally paid within...
-
Anita Maxwell Company expects the following for 2007: Net cash provided by operating activities of $150,000 Net cash provided by financing activities of $60,000 Net cash used for investing...
-
According to ightstats.com, American Airlines ights from Dallas to Chicago are on time 80% of the time. Suppose 15 ights are randomly selected, and the number of on-time ights is recorded. (a)...
-
The following are comparative balance sheets for Bridgeport Company. Bridgeport Company Comparative Balance Sheets December 31 2025 2024 Assets Cash $108,800 $35,200 Accounts receivable 140,800...
-
Consider Table 6.19, from a study of nonmetastatic osteosarcoma. The response is whether the subject achieved a three-year disease-free interval. a. Show that each predictor has a significant effect...
-
How can a molecular formula be obtained from an empirical formula? What additional information is required?
-
What is combustion analysis? What is it used for?
-
A firm has a long-term debtequity ratio of .4. Shareholders equity is $1 million. Current assets are $200,000, and the current ratio is 2. The only current liabilities are notes payable. What is the...
-
If a computer passed the Turing Test, what would that prove? What conditions would you want to be sure had been observed in setting up the test?
-
What is global account management? What competences are required and do they differ from those required of the key account manager?
-
What would be the implications of Focus Wickes moving to 50 per cent own label merchandise within the next year from the viewpoints of: end customers sales staff suppliers?
-
Explain why Depth-First Search Iterative Deepening is reasonably efficient. Why might it be preferable to use DFID rather than depth-first search?
-
What are the primary characteristics of (a) sequential and (b) random processing?
-
How does S&OP differ from strategic capacity planning? From detailed planning and control? What role does S&OP play in the overall planning activities of an organization?
-
If the amplifier indicated by the box input impedance of oo, which of the following statements are true ? has an open loop gain as well as Feedback factor (\beta = 1/ R_1\) The feedback is voltage...
-
Ascorbic acid (vitamin C) does not contain a traditional carboxylic acid group, but it is, nevertheless, still fairly acidic (pKa = 4.2). Identify the acidic proton, and explain your choice using...
-
In the following compound two protons are clearly identified. Determine which of the two is more acidic. After comparing the conjugate bases, you should get stuck on the following question: Is it...
-
Identify the most acidic proton in each of the following compounds and explain your choice: (a) (b) 0-H F3C 0-H CI. CI
-
Gladstone Company tracks the number of units purchased and sold throughout each accounting period but applies its inventory costing method at the end of each period, as if it uses a periodic...
-
A company is negotiating wages with its union. The firm's profits are E = 10 where is the salary. The objective function of the union union is U = 10 (^2) 5. The rules of wage bargaining are as...
-
As shown in the figure below, a rectangular wire of side lengths a and b, and carrying a current 13 is placed in between two infinitely-long wires, carrying currents / and 12, respectively. All...

Study smarter with the SolutionInn App


