Calculate the change in entropy that occurs in the system when 45.0 g of acetone (C 3
Question:
Calculate the change in entropy that occurs in the system when 45.0 g of acetone (C3H6O) freezes at its melting point (-94.8 °C).
See Table 12.9 for heats of fusion.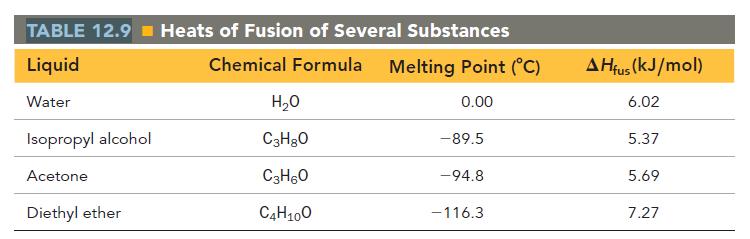
Transcribed Image Text:
TABLE 12.9 Heats of Fusion of Several Substances Liquid Chemical Formula Melting Point (°C) Water 0.00 Isopropyl alcohol Acetone Diethyl ether H₂O C3H8O C3H6O C4H10O - 89.5 -94.8 -116.3 AHfus (kJ/mol) 6.02 5.37 5.69 7.27
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the change in entropy that occurs in the system when 1.00 mole of isopropyl alcohol (C 3 H 8 O) melts at its melting point (-89.5 C). See Table 12.9 for heats of fusion. TABLE 12.9 Heats of...
-
Calculate the change in entropy that occurs in the system when 55.0 g of water vaporizes from a liquid to a gas at its boiling point (100.0 C). See Table 12.7 for heats of vaporization. TABLE 12.7...
-
Calculate the change in entropy that occurs when 18.02 g of ice at 210.0oC is placed in 54.05 g of water at 100.0oC in a perfectly insulated vessel. Assume that the molar heat capacities for H2O(s)...
-
The largest government expenditure for Japan & the US is... a) military / defense b) transfer payments c) education / healthcare d) infrastructure
-
What are some problems created by language and the ability to comprehend the questions in collecting primary data? How can a foreign market researcher overcome these difficulties?
-
For the following project, (a). List all predecessors of task 5. (b). List all predecessors of task 4. (c). List all predecessors of the network finish(F). 5 in 2.
-
In 2014, political consulting firm Cambridge Analytica developed an app designed to create digital profiles of individuals via their information. Cambridge Analytica collected the data by inviting...
-
Clarence Weatherspoon, a super salesman contemplating retirement on his fifty-fifth birthday, decides to create a fund on an 8% basis that will enable him to withdraw $20,000 per year on June 30,...
-
the tuition fee for maths tuition for john is expected to be 6500 per year if he attend tuition for 4 year starting from 4 years from now how much does he have to have in her account which yields 6...
-
Without doing any calculations, determine the sign of S sys for each chemical reaction. a. Mg(s) + Cl(g) MgCl(s) b. 2 HS(g) + 3 O(g) 2 HO(g) + 2 SO(g) c. 203(g) d. HCI(g) + NH3(g) NH4Cl(s) 3 O(g)
-
Calculate the change in entropy that occurs in the system when 1.00 mole of diethyl ether (C 4 H 10 O) condenses from a gas to a liquid at its normal boiling point (34.6 C). See Table 12.7 for heats...
-
A survey of athletic trainers asked what modalities (treatment methods such as ice, whirlpool, ultrasound, or exercise) they commonly use to treat injuries. Respondents were each asked to list three...
-
Assume a large local manufacturer calls you for some economic advice. Their workforce is predominantly male and average age is 4 2 , and the prevalence of heart disease and stroke in their population...
-
McDonald's When Steve Easterbrook took over as CEO at McDonald's, the changes were dramatic and swift. Gone was the "family" atmosphere and in was the "team." (You can't choose your family members,...
-
A body of mass m-2kg is initially at position, r--(1+2)) and moves with an Initial velocity, v, -(-4+21) and an acceleration, a=(1+21) where g-10m/s and r-3s are two constant parameters having...
-
Critical path can be used by the Project Manager to focus on which project activities can NOT slip or the project will slip and use to calculate Float focus on which project activities can not slip...
-
Thinking strategically is critical for any organization to compete successfully and build the necessary competitive advantage for sustained superior performance. Managers and business leaders will be...
-
Discuss the advantages of some of the current technologies described in our reading this week including VoIP, Unified Communications, Virtual Private Networks, Wi-Fi and wireless technologies like...
-
Chicago Company sold merchandise to a customer for $1,500 cash in a state with a 6% sales tax rate. The total amount of cash collected from the customer was $558. $600. $642. $636. Nevada Company...
-
With the previous problem in mind show that the inverse transform of
-
Show that if Æ(x) is real and even, its transform is real and even. Start with Eq. (11.5), use the Euler formula from Section 2.5, and assume that Æ(x) has both a real and an imaginary...
-
Given that F{(x)} = F() and F{h(x)} = H(), if and b are constants, determine F{(x) + bh(x)}.
-
This speech is a Fact/Value speech outline Need to include introduction, body, conclusions and sources at 5. Please make sure it's a proper outline and read & follow all the instructions below....
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
Using the policies and procedures for Gold Catering: Explain the different procedures involved in recording bad debts and doubtful debts: Why is there a difference between bad debt and doubtful debt?...

Study smarter with the SolutionInn App


