Determine whether each redox reaction is spontaneous. (a) Fe(s) + Mg2+ (aq) (b) Fe(s) + Pb+ (aq)
Question:
Determine whether each redox reaction is spontaneous.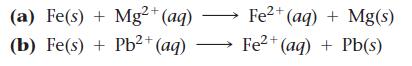
Transcribed Image Text:
(a) Fe(s) + Mg2+ (aq) (b) Fe(s) + Pb²+ (aq) Fe2+ (aq) + Mg(s) 2+ Fe²+ (aq) + Pb(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Fes Mg2 aq Fe aq Mgs This reaction involves the oxidation of Fe Fes Fe aq 2e w...View the full answer

Answered By

Rashul Chutani
I have been associated with the area of Computer Science for long. At my university, I have taught students various Computer Science Courses like Data Structures, Algorithms, Theory of Computation, Digital Logic, System Design, and Machine Learning. I also write answers to questions posted by students in the area of and around Computer Science.
I am highly fortunate to receive great feedback on my teaching skills that keeps me motivated. Once a student sent me an email stating that I had explained to him a concept better than his professor did.
I believe in the fact that "Teaching is the best way to learn". I am highly fascinated by the way technology nowadays is solving real-world problems and try to contribute my bit to the same.
Besides tutoring, I am a researcher at the Indian Institute of Technology. My present works are in the area of Text Summarization and Signal and Systems.
Some of my achievements include clearing JEE Advanced with an All India Rank of 306 out of 1.5 million contesting candidates and being the Department Ranker 1 at my University in the Department of Computer Science and Engineering.
I look forward to providing the best Tutoring Experience I can, to the student I teach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ca+ (aq) + Zn(s) b. 2 Ag+ (aq) + Ni(s) c. Fe(s) + Mn+ (aq) d. 2 Al(s) + 3 Pb+ (aq) Ca(s) + Zn+ (aq) 2 Ag(s) +...
-
Determine whether each redox reaction occurs spontaneously in the forward direction. a. Ni(s) + Zn+ (aq) b. Ni(s) + Pb+ (aq) c. Al(s) + 3 Ag+ (aq) d. Pb(s) + Mn+ (aq) 2+ Ni+(aq) + Zn(s) Ni+(aq) +...
-
Determine whether each reaction is a redox reaction. For each redox reaction, identify the oxidizing agent and the reducing agent. a. Al(s) + 3 Ag+ (aq) b. SO3(g) + HO(1) c. Ba(s) + Cl(g) d. Mg(s) +...
-
Find the exact value of sin(x - y) if sin(x) = 3T 3T T
-
What are some of the benefits of a centralized database architecture? What are some of the difficulties in moving from multiple databases or files to a centralized database structure?
-
Four particles at the corners of a square with side length L = 2 m are connected by massless rods (Figure). The masses of the particles are m 1 = m 3 = 3 kg and m 2 = m4 = 4 kg. Find the moment of...
-
Plaintiffs purchased stock warrants (rights to purchase) for blocks of Osborne Computer Corp., the manufacturer of the first mass-market portable personal computer. Because of inability to produce a...
-
Bridgette Keyes is a licensed dentist. During the first month of the operation of her business, the following events and transactions occurred. April 1 Invested $20,000 cash in her business. 1 Hired...
-
A manufacturing company reports the following information. Raw materials inventory, ending Raw materials used Current Year $ 169,500 2,160,000 1 Year Ago 2 Years Ago $ 190,500 2,522,000 $ 197,500...
-
What reactant types give rise to gas-evolution reactions?
-
What is a gas-evolution reaction? Give an example.
-
Corporate sustainability reports vary greatly across companies and industries. Select two companies that interest you and conduct an online search to find their corporate sustainability report. (If...
-
Customers moving up future purchases to the present is market growth. stealing share. forward selling. forward buying.
-
Devise appropriate discounting schemes for a supply chain.
-
Customers substituting the firms product for a competitors product is known as market growth. stealing share. forward selling. forward buying.
-
In most instances of differential pricing, demand from the segment paying the lower price arises earlier in time than demand from the segment paying the higher price. arises later in time than demand...
-
Pricing decisions based only on revenue considerations often result in a decrease in overall profitability. an increase in overall profitability. a decrease in overall revenue. a decrease in supply...
-
Last year, Owens, Inc., reported bad debt expense of $150,000. During the current year, Owens discovered that its bad debt expense for last year should have been $175,000. Owens retained earnings had...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
If Saturn could fit in a bathtub, would it float? Use the planetary data in Appendix A to calculate the density (mass/volume) of Earth, Mars, Jupiter, and Saturn. What are the differences? Can you...
-
A person stretches a rope of length 55 m between the top of a building to the base of a tree 25 m away (Fig. P1.71). How tall is the building? Figure P1.71 ? -Rope +25 m|
-
At room temperature, 1.0 g of water has a volume of 1.0 cm 3 . (a) What is the approximate volume of one water molecule? (b) The human body is composed mainly of water. Assuming for simplicity your...
-
Scrappers Supplies tracks the number of units purchased and sold throughout each accounting period but applies its inventory costing method at the end of each period, as if it uses a periodic...
-
Remo Company and Angelo Incorporated are separate companies that operate in the same industry. Following are variable costing income statements for the two companies showing their different cost...
-
The first production department of Stone Incorporated reports the following for April. Direct Materials Conversion Units Beginning work in process inventory 69,000 Percent Complete sex Percent...

Study smarter with the SolutionInn App


