Iodine is made by the following reaction (a) Name the two reactants. (b) If you wish to
Question:
Iodine is made by the following reaction 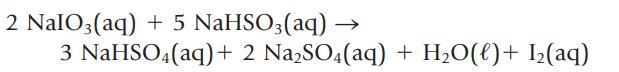
(a) Name the two reactants.
(b) If you wish to prepare 1.00 kg of I2, what masses of NaIO3 and NaHSO3 are required?
(c) What is the theoretical yield of I2 if you mixed 15.0 g of NaIO3 with 125 mL of 0.853 M NaHSO3?
Transcribed Image Text:
2 NalO3(aq) + 5 NaHSO3(aq) 3 NaHSO4(aq) + 2 Na₂SO4(aq) + H₂O(l) + I₂(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a The two reactants in the given chemical reaction are Sodium Iodate NaIO3 Sodium Bisulfite NaHSO3 b To prepare 100 kg of I2 iodine you need to determ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The mass of the uniform slender rod is 3 kg. Determine the position x for the 1.2-kg slider such that the system period is 1 s. Assume small oscillations about the horizontal equilibrium position...
-
The uniform 5-kg rod rests on the ground at B and against the wall at A. If the rod does not slip at B, determine the smallest coefficient of static friction at A which will prevent slipping at A. X...
-
Determine the horizontal and vertical components of force at pins B and C. The suspended cylinder has a mass of 75 kg. 0.3 m B 1.5 m A 2 m 0.5 m
-
Danbury Inc is a resident Canadian corporation with worldwide operations. Canadian operations resulted in taxable income of $ 1 . 1 0 million and non - Canadian operations resulted in taxable income...
-
Find an example of successful TQM implementation.
-
What happened to the marginal utility derived from food consumption as people responded to lower food prices by purchasing more food?
-
Water flows over a flat surface at \(4 \mathrm{ft} / \mathrm{s}\), as shown in Fig. P6.42. A pump draws off water through a narrow slit at a volume rate of \(0.1 \mathrm{ft}^{3} / \mathrm{s}\) per...
-
We might expect credit card purchases to differ from cash purchases at the same store. Table P-15 contains daily gross sales and items sold for cash purchases and daily gross sales and items sold for...
-
A crane lifts a load with a force of 1000 N over a distance of 10 meters in 20 seconds. What is the average power generated by the crane?
-
Boron forms a series of compounds with hydrogen, all with the general formula B x H y . If 0.148 g of one of these compounds gives 0.422 g of B 2 O 3 when burned in excess O 2 , what is its empirical...
-
Sodium azide, an explosive chemical used in automobile airbags, is made by the following reaction: If you combine 15.0 g of NaNO 3 with 15.0 g of NaNH 2 , what mass of NaN 3 is produced? NaNO3 + 3...
-
Follow these steps to organize employment information. 1. Review the employment ads in a local or regional newspaper. 2. Collect information on at least 10 job titles plus the skills and education...
-
Ace Co. issued 1,000 shares of its $10 par value common stock for $15 per share in cash How should this transaction be reported in Ace's statement of cash flows for the year of issuance?
-
.A man shows up at the emergency room at a hospital. You are called to consult on this case. The man does not know his own name. He is unable to identify what city he lives in, and is not sure how he...
-
You have probably noticed in your educational career that some people are very good atremembering facts and therefore do well at tests that require memorization. Other students, onthe other hand,...
-
Maximize the product of two numbers if their sum must be 14. The two numbers are (Use a comma to separate answers as needed.)
-
3. A research study shows that men who have facial tattoos earn less than men who do not have tattoos on their faces. Would the results of this study then indicate that employers discriminate against...
-
How is the purchase of a held-to-maturity debt security at face value recorded?
-
Briefly discuss the implications of the financial statement presentation project for the reporting of stockholders equity.
-
The compound below is believed to be a wasp pheromone. Draw the major product formed when this compound is hydrolyzed in aqueous acid:
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OMe Meo Meo Br Br
-
In a calorimetric study, the temperature of the system rises to 325 K before returning to its initial temperature of 298 K. Why doesnt this temperature rise affect your measurement of H o R at 298 K?
-
Your team's organisation, Awesome IT has been engaged by SAIBT to explore accessible learning requirements for SAIBT's online student newsletter platform. You are invited to explore a range of...
-
Bob has to send digitally signed messages (documents) to his banks. The digital signature scheme used by the banks is based on RSA. It is the simple scheme, discussed initially in the lectures, which...
-
Operational security (OPSEC) is a form of risk management that challenges managers in an organization to think like an adversary (Zhang, 2020). OPSEC is used to protect organizational assets,...

Study smarter with the SolutionInn App


