Discuss the effect of temperature change on the spontaneity of the following reactions at 1 atm. (a)
Question:
Discuss the effect of temperature change on the spontaneity of the following reactions at 1 atm.
(a) 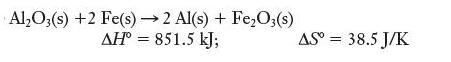
(b) 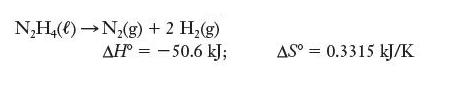
(c) 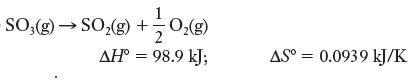
Transcribed Image Text:
Al₂O3(s) +2 Fe(s)→2 Al(s) + Fe₂O3(s) ΔΗ° = 851.5 kJ; AS = 38.5 J/K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To determine the effect of temperature change on the spontaneity of the given reactions we can use t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Consider the following lattice structure of generalization and specialization A X B Y C
-
Determine whether the quadrilateral is a trapezoid. Explain. A D B J C
-
Discuss the effect of temperature change on the spontaneity of the following reactions at 1 atm. (a) Al2O3(s) + 2Fe(s) > 2Al(s) + Fe2O3(s) [ H = +851.5 kJ; S = +38.5 J/K] (b) N2H4(l) > N2(g) +...
-
Someone offered the investment options to Hendry on January 1, 2023: 1. Hendry has to save up to 5 times the initial deposit of US $ 150,000/year. 2. The savings cannot be taken for 20 years until...
-
What is an asset retirement obligation? What is the proper accounting for an asset retirement obligation?
-
The following errors took place in journalizing and posting transactions: a. Utilities expense of $7,300 paid for the current month was recorded as a debit to Miscellaneous Expense and a credit to...
-
How do stakeholder groups relevant to environmental management accounting and environmentally induced financial accounting differ?
-
Your roommate, Matt Mikan, contends that accounting contributes to most of the steps in managements decision- making process. Is your roommate correct? Explain.
-
Explain what step-rate integration is and illustrate how it might work for a defined benefit plan. What does "vesting" mean, and what role does vesting play in the history of pension standards...
-
The combustion of acetylene was used in welders torches for many years because it produces a very hot flame: (a) Use data in Appendix E to calculate S for this reaction. (b) Calculate G and show that...
-
There is another free energy state function, the Helmholtz free energy (A), defined as A = E TS. Comparing this to the definition of G, we see that internal energy has replaced enthalpy in the...
-
Repeat Problem 11.26 assuming the turbine has an isentropic efficiency of 85%.
-
Compare and contrast the two conflicts in your own words. 1. My team was disrupted by a colleague's behavior at work recently, and I had to confront him about the issue. As a result of the conflict,...
-
Subtract. 17c-2 3c 4c 4c Simplify your answer as much as possible.
-
What is the time complexity of the following function named func? 1 int func(int n) { //n>0 if (n < 2) { return 1; } else if (n%2 == ) { AWNH 3 4 567 6 8 9} return func (n/3); } else { } return...
-
As you work to become an educator, it is important to think about and be able to explain how you will respect students with differing personal backgrounds and various skills, abilities, perspectives,...
-
Justin is 16. Justin went through puberty early and appears older than he really is. One day after school, Justin gets in a fight with another teenager. Justin is punched in the mouth and his teeth...
-
Are random arrivals at a shoe store at the local mall Poisson distributed? Suppose a mall employee researches this question by gathering data for arrivals during one-minute intervals on a weekday...
-
Cable Corporation is 60% owned by Anna and 40% owned by Jim, who are unrelated. It has noncash assets, which it sells to an unrelated purchaser for $100,000 in cash and $900,000 in installment...
-
What observable is used to measure the viscosity of a gas or liquid?
-
In describing viscosity, what system quantity was transported? What is the expression for viscosity in terms of particle parameters derived from gas kinetic theory?
-
At 30 km above the Earths surface (roughly in the middle of the stratosphere), the pressure is roughly 0.013 atm and the gas density is 3.74 Ã 1023 molecules/m3. Assuming N2 is representative...
-
What is BYD Global's revenue analysis from 2018-2022 in terms of revenue earned and profit margin based on the chart 1. Operating revenue Less: Operating costs Tax and surcharge Selling expenses...
-
Troy Engines, Limited, manufactures a variety of engines for use in heavy equipment. The company has always produced all of the necessary parts for its engines, including all of the carburetors. An...
-
1. Company Value Audit (Make it for Apple) A) What are the returns on investments for these brands? B)How much market share does the brand have? C) Is there a price premium advantage for products? D)...

Study smarter with the SolutionInn App


