Which of the following compounds would you predict to be soluble in water at room temperature? (a)
Question:
Which of the following compounds would you predict to be soluble in water at room temperature?
(a) KClO3,
(b) CaCO3,
(c) BaSO4,
(d) KMnO4
Strategy Solubility guidelines for common ions are given in Table 3.1. So we will identify the ions in each compound and consult the table as needed to determine the solubilities.
Table 3.1
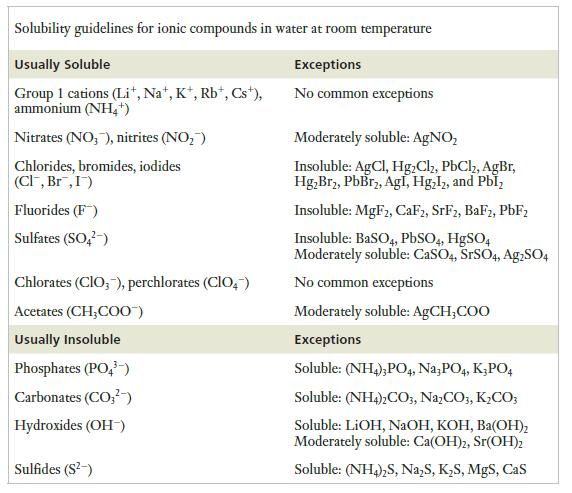
Transcribed Image Text:
Solubility guidelines for ionic compounds in water at room temperature Usually Soluble Exceptions No common exceptions Group 1 cations (Li+, Na+, K+, Rb+, Cst), ammonium (NH4+) Nitrates (NO3), nitrites (NO₂) Chlorides, bromides, iodides (Cl, Br, I) Fluorides (F) Sulfates (SO²) Chlorates (ClO3), perchlorates (CIO) Acetates (CH₂COO) Usually Insoluble Phosphates (PO4³-) Carbonates (CO²) Hydroxides (OH-) Sulfides (S²-) Moderately soluble: AgNO₂ Insoluble: AgCl, Hg2Cl2, PbCl2, AgBr, Hg₂Br₂, PbBr₂, Agl, Hg₂12, and Pbl₂ Insoluble: MgF2, CaF2, SrF2, BaF2, PbF₂ Insoluble: BaSO4, PbSO4, HgSO4 Moderately soluble: CaSO4, SrSO4, Ag2SO4 No common exceptions Moderately soluble: AgCH,COO Exceptions Soluble: (NH4), PO4, Na3PO4, K3PO4 Soluble: (NH4)2CO3, Na₂CO3,K₂CO3 Soluble: LiOH, NaOH, KOH, Ba(OH)₂ Moderately soluble: Ca(OH)2, Sr(OH)2 Soluble: (NH4)₂S, Na₂S, K₂S, MgS, CaS
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a KClO 3 is potassium chlorate From the solubility guidelines in Table 31 we see that compound containing K and ClO 3 tend to be soluble and that no c...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds would you expect to be most acidic? Justify your choice.
-
Which of the following compounds would you expect to be more reactive in an SN2 reaction? CH3 CH3Br CH3H
-
You have a net income of $40 000 per year. Your expenses include the following: Rent: $800 per month Insurance: $225 per semi-annually Car Payment: $315 per month Car Expenses: $1 000 per year ...
-
Marian Kirk wishes to select the better of two 10-year annuities, C and D. Annuity C is an ordinary annuity of $2,500 per year for 10 years. Annuity D is an annuity due of $2,200 per year for 10...
-
Kase Company can invest in each of three cheese-making projects: C1, C2, and C3. Each project requires an initial investment of $190,000 and would yield the following annual cash flows. (1) Assuming...
-
Mega Millions is a multistate lottery in which players try to guess the numbers that will turn up in a drawing of numbered balls. One of the balls drawn is the Mega Ball. Matching the number drawn on...
-
In Section 5.5, we showed the following two-person, zero-sum game had a mixed strategy: a. Use dominance to reduce the game to a 2 2 game. Which strategies are dominated? b. Determine the optimal...
-
Describe differing types of currency exchange rate financial risk exposures. Discuss the methods a company may use to reduce the potentially significant negative impact of these different types of...
-
Propane, C 3 H 8 , is used as a fuel in many applications, including gas barbecue grills. Because of its widespread use, extensive research is underway to develop ways to produce propane from...
-
Name each of the following compounds: (a) MgCl 2 , (b) Fe (NO 3 ) 2 , (c) Na 2 SO 4 , (d) Ca (OH) 2 , (e) FeSO 4
-
For the following, assume that Plants A and B have the following marginal costs for reducing emissions: (a) The government tells both plants to reduce three units of output. How much does this...
-
2.) For the rectangle below, If side x = 5.675 ft and side y = 2.3 ft, calculate: X a.) The perimeter of the rectangle. b.) The area of the rectangle. y
-
Make a program using the javascript that takes a radius as input. Using these input, solve for the circumference and print it on the next line. Assume PI is 3.14. Input 1. One line containing an...
-
Towne Center Bonds pay 12% coupon and have a maturity of 10 years, while Village Market Bonds pay 6% coupon and have a maturity of 20 years. How do coupon and maturity impact the volatility of Towne...
-
Bolsters food balance sheet shows a total of 25 million long term debt with a coupon rate of 8.50 the yield to maturity on this debt is 8.00 and the debt has a total current market value of 27...
-
Albert who is a resident of Canada attended the University of Toronto on a full-time basis for 8 months during 2023. While there he earned employment income for tax purposes of $19,000 for 2023. He...
-
Elaine Shumate has been working for GSM, a pharmaceutical research company, for more than seven years. It is her first job since finishing her graduate work in molecular biology, and her performance...
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
In the reversible adiabatic expansion of 1.75 mol of an ideal gas from an initial temperature of 27.0C, the work done on the surroundings is 1300. J. If C V ,m = 3/2R, calculate q, w, U, and H.
-
For a given set of conditions, the fugacity of a gas is greater than the pressure. What does this tell you about the interaction between the molecules of the gas?
-
Predict the major product of the reaction between 1-butanol and: (a) PBr 3 (b) SOCl 2 , py (c) HCl, ZnCl 2 (d) H 2 SO 4 , heat (e) PCC, CH 2 Cl 2 (f ) Na 2 Cr 2 O 7 , H 2 SO 4 , H 2 O (g) Li (h) NaH...
-
This case showcases Singhania and Partners, a full-service national law firm in India, and analyzes their HR practices. Since India's economic liberalization which began in the 1990s, the Indian...
-
"Cell phones and their applications are developing at a very fast pace. These technological developments have major influences, posing opportunities as well as threats to organisations. With...
-
MaBelle Corporation, which uses straight-line depreciation and amortization, incurred the following costs in 2026: Cost of R&D equipment with a useful life of 4years in R&D projects (no salvage)...

Study smarter with the SolutionInn App


