Measurements of conductivity of solutions of two acids, A and B, produced the following data. Characterize each
Question:
Measurements of conductivity of solutions of two acids, A and B, produced the following data. Characterize each acid as strong or weak.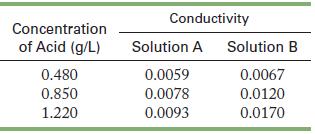
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
A is a ...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Two 0.10-mol samples of the hypothetical monoprotic acids HA(aq) and HB(aq) are used to prepare 1.0-L stock solutions of each acid. a. Write the chemical reactions for these acids in water. What are...
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HF b. HCHO c. HSO4 d. HCO3
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HNO3 b. HCI c. HBr d. HSO3
-
Why is it that I got 0 Row with nothing showing up? Is there a flow in my logic? Using AdventureWorks DW2017, list all the Canadian customers who have spent more than $5,000 total. Show Customerkey,...
-
The human brain is especially sensitive to elevated temperatures. The cool blood in the veins leaving the face and neck and returning to the heart may contribute to thermal regulation of the brain by...
-
Electricity and process heat requirements of a manufacturing facility are to be met by a cogeneration plant consisting of a gas turbine and a heat exchanger for steam production. The plant operates...
-
Probability affects decisions when the weather is forecast, when medications are selected, and even when players are selected for professional sports teams. Although intuition is often used for...
-
Voss Company acquired patent rights on January 6, 2011, for $480,000. The patent has a useful life equal to its legal life of eight years. On January 3, 2014, Voss successfully defended the patent in...
-
A. Calculate the cross-arc elasticity in the following situation. (mathematical method) Price X Amount Y $10 70 $13 100 Based on the above result, consumer response regarding products can be...
-
Assuming that the conductivity of an acid solution is proportional to the concentration of H 3 O + , sketch plots of conductivity versus concentration for HCl and HF over the 0- to 0.020 M...
-
A solution is prepared by dissolving 0.121 g uric acid, C 5 H 3 N 4 O 3 H (molar mass = 168 g/mol), and diluting to make exactly 10 mL of solution. Each uric acid molecule has only one hydrogen ion...
-
For the following exercises, make a table of values for each set of parametric equations, graph the equations, and include an orientation; then write the Cartesian equation. A ball is launched with...
-
Consider a massive string of constant linear mass density and length L whose endpoints are fixed at A (x = 0, z = z0) and B (x = a, z = z1). The string lies in the vertical plane (x, z), and it is...
-
Show that the derivative of an integral is given by By generalizing to three dimensions show that the material derivative of an integral of a fluid property is given by where the surface integral ( s...
-
Consider a pendulum of length l and mass m2 hanging on a point of mass m1 that moves horizontally without friction on a rail.We note x the abscissa of m1 and the angle with the vertical direction....
-
The problem of three coupled oscillators is treated in analogous manner as the twobody case of Sect. (4.3) with the Jacobi variables. The Hamiltonian is The canonical transformation (Jacobi...
-
Name a past conflict and how this conflict affected the business.
-
Refer to the background information in Exercise 17-60 for the Destin Company. One of the quality improvements management instituted recently was the use of statistical control charts. Over the most...
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
Design a multiplier for 16-bit binary integers. Use a design similar to Figures 4-33 and 4-34. (a) Draw the block diagram. Add a counter to the control circuit to count the number of shifts. (b) Draw...
-
The block diagram for an elevator controller for a building with two floors is shown in the following diagram. The inputs FB 1 and FB 2 are floor buttons in the elevator. The inputs CALL 1 and CALL 2...
-
A century ago an entirely new group of elements, the inert gases, was discovered. Is it possible that, in the future, another as yet unknown group of the periodic table might be found?
-
Consider an investor that bought the newly-issued Starlight bond 3 years ago for $1,000. If the bond is currently valued at $980, what is their total investment return over the 3-year holding period...
-
In September 2023, suppose FedEx had the following characteristics: Stock price = $250/share Shares outstanding = 256 million Net Debt (debt - cash) = $13.7 billion Equity beta = 1.40 i. Using this...
-
1. Revenue Collection: The Urban Institute Backgrounder reading, State and Local Revenues reports total revenues for U.S. local governments in 2020. The reading by Erin Duffin (Statista)reports total...

Study smarter with the SolutionInn App


