Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid
Question:
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions.
(a) Hydroxylammonium ion
(b) Ammonium ion
Table 15.6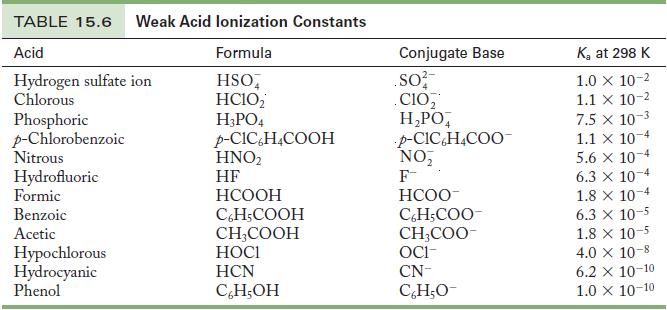
Table 15.8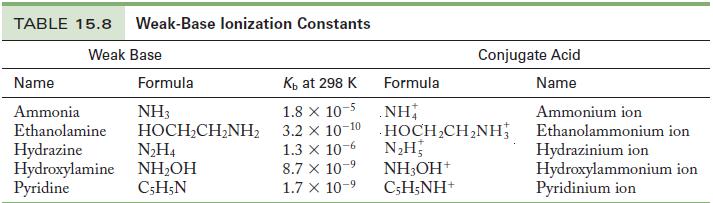
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a NH3OH HONHOH H3O Kw ...View the full answer

Answered By

Rustia Melrod
I am a retired teacher with 6 years of experience teaching various science subjects to high school students and undergraduate students. This background enables me to be able to help tutor students who are struggling with the science of business component of their education. Teaching difficult subjects has definitely taught me patience. There is no greater joy for me than to patiently guide a student to the correct answer. When a student has that "aha!" moment, all my efforts are worth it.
The Common Core standards are a useful yardstick for measuring how well students are doing. My students consistently met or exceeded the Common Core standards for science. I believe in working with each student's individual learning styles to help them understand the material. If students were struggling with a concept, I would figure out a different way to teach or apply that concept. I was voted Teacher of the Year six times in my career. I also won an award for Innovative Teaching Style at the 2011 National Teaching Conference.
4.90+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Is the criterion 6 3CO 2 ) 2 (aq) is found to be 9.11. However, the contribution to the pH from the autoprotolysis of water was ignored. Repeat the calculation of the pH of this solution, taking into...
-
Calcium acetate, Ca(CH 3 CO 2 ) 2 (aq), is used to treat patients with a kidney disease that results in high levels of phosphate ions in the blood. The calcium binds to the phosphates so that they...
-
Although there are extensive tables available for the pK a of weak acids, you might be dealing with an unknown acid or a known acid at an unlisted temperature. You could then use a procedure like...
-
Explain what quality management knowledge and skills an auditor should possess when auditing
-
Consider the influence of a finite sheet thickness in Example 11.2, when there are 40 gaps. (a) Determine the exterior dimension, L, of the heat exchanger core for a sheet thickness of t = 0.8 mm for...
-
Consider a Carnot cycle executed in a closed system with 0.003 kg of air. The temperature limits of the cycle are 300 and 900 K, and the minimum and maximum pressures that occur during the cycle are...
-
A code consists of two distinct letters followed by three digits. The last digit cannot be 0 or 1. What is the probability of guessing the security code on the first try? Use counting principles to...
-
Sonimad Sawmill Inc. (SSI) purchases logs from independent timber contractors and processes the logs into three types of lumber products: Studs for residential buildings (walls, ceilings). Decorative...
-
Fanshawe college Course Code : MGMT 6147 : Trends in Management & Leadership Module & Case: #10: Motivated reasoning, leadership, and team performance. 1) Please provide a brief summary of the case....
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions. (a) Pyridinium ion (b) Hydrazinium ion Table 15.6 Table 15.8
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the base ionization constant for the following ions. (a) Chlorite ion (b) Fluoride ion Table 15.6 Table 15.8
-
Given the following information about Elkridge Sporting Goods, Inc., construct a balance sheet for June 30, 2011. On that date the firm had cash and marketable securities of $25,135, accounts...
-
Conduct an audit of your mediated interpersonal and masspersonal messages. a. Using the format in Figure 2.2, plot the scope of your mediated interpersonal and masspersonal communication. b. Describe...
-
In this chapter, you learned about three main emphases for account planners: developing consumer insights, formulating a strategy, and evaluating the effectiveness of the message. Now think about...
-
The At Work box in Section 3.4 describes how organizations have the properties of a culture. Select a place of business where you have worked and describe in a few sentences its organizational...
-
Which of these best describes the business use of social networks? a. Businesses use private social networks but not public networks. b. Businesses use private and public social networks. c....
-
As student representatives, your colleagues have suggested the following six presentation purposes for a staffstudent conference. Summarize your choice, giving reasons: a. To show the audience that...
-
Discuss the difference between goalpost conformance and absolute quality conformance.
-
A woman at a point A on the shore of a circular lake with radius 2 mi wants to arrive at the point C diametrically opposite on the other side of the lake in the shortest possible A time. She can walk...
-
Write a Verilog module that describes a 16-bit serial-in, serial-out shift register with inputs SI (serial input), EN (enable), CK (clock, shifts on rising edge), and a serial output (SO).
-
A description of a 74194 4-bit bidirectional shift register follows. The CLRb input is asynchronous and active low and overrides all the other control inputs. All other state changes occur following...
-
A synchronous (4-bit) up/down decade counter with output Q works as follows: All state changes occur on the rising edge of the CLK input, except the synchronous clear (CLR). When CLR = 0, the counter...
-
Consider the truss below, secured at the nodes via clevis joints. (a) Sketch a FBD for elements AC, CE, BD, and the Clevis Pin in Joint D. Determine all reaction forces. (b) Generate the Shear-Moment...
-
a) Suppose f: R Z, where f(x) = x + | i. Graph the function f(x). ii. If A = {x: 1 x 3}, find f(A). iii. Find f({0}) iv. Find f({-1,0,1)}) b) Let the functions f, g and h be defined as follows: f: R...
-
Assume that Sigma Group has a market equity capitalisation of $10.8 billion and its enterprise value is $14.4 billion. The company's equity holders require a return of 10% and the debtholders a...

Study smarter with the SolutionInn App


