Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid
Question:
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions.
(a) Pyridinium ion
(b) Hydrazinium ion
Table 15.6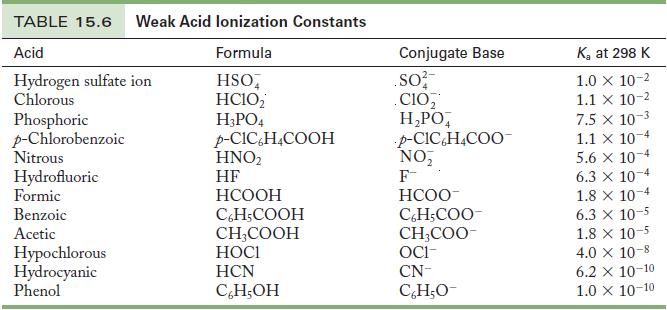
Table 15.8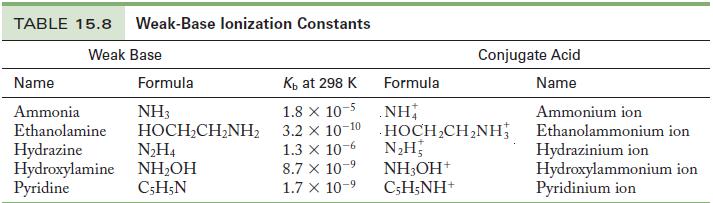
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Answer Acid Ionization Constants of Pyridinium and Hydrazinium Ions a Pyridinium Ion C5H5NH Pyridini...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Is the criterion 6 3CO 2 ) 2 (aq) is found to be 9.11. However, the contribution to the pH from the autoprotolysis of water was ignored. Repeat the calculation of the pH of this solution, taking into...
-
Calcium acetate, Ca(CH 3 CO 2 ) 2 (aq), is used to treat patients with a kidney disease that results in high levels of phosphate ions in the blood. The calcium binds to the phosphates so that they...
-
Although there are extensive tables available for the pK a of weak acids, you might be dealing with an unknown acid or a known acid at an unlisted temperature. You could then use a procedure like...
-
A. What is Alpha One's NOPAT? Why does NOPAT differ from the earnings after taxes? B. Estimate the effective before-tax cost of the long-term debt. c. Estimate the effective after-tax cost of the...
-
Hot exhaust gases are used in a shell-and-tube exchanger to heat 2.5 kg/s of water from 35 to 85C. The gases assumed to have the properties of air, enter at 200C and leave at 93C. The overall heat...
-
Define the following terms related to reciprocating engines: stroke, bore, top dead center, and clearance volume.
-
A batch of 100 mobile phones contains 5 smartphones. What is the probability that a sample of three phones will have (a) no smartphones? (b) all smartphones? (c) at least one smartphone? (d) at least...
-
On August 3, 2010, the date of incorporation, the Quinn Company accepts separate subscriptions for 1,000 shares of $100 par preferred stock at $104 per share and 9,000 shares of no-par,...
-
Non-current liabilities Bonds payable Equity Share capital-ordinary, 1.00 par, authorized 400,000 shares, issued 295,500 Share premium-ordinary Retained earnings 505,500 295,500 165,500 ? Prepare a...
-
Find the value of K b for the conjugate base of the following organic acids. (a) Picric acid used in the manufacture of explosives; K a = 0.16 (b) Trichloroacetic acid used in the treatment of warts;...
-
Write the chemical equation and use the data in Tables 15.6 and 15.8 to calculate the acid ionization constant for the following ions. (a) Hydroxylammonium ion (b) Ammonium ion Table 15.6 Table 15.8
-
Based on the problem statement of Example 14.20, design a four-stage diafiltration section to take the 55 wt% concentrate from Section 1 and achieve the desired 85 wt% concentrate, thus eliminating...
-
Identify one culture with which you currently interact or could interact in the future. Collect information on communication rules and norms in that culture through library/internet research and...
-
With a group of classmates, compile a list of reference groups you use to define your self-concepts. You can recognize them by answering several questions: a. Select one area in which you compare...
-
Improve your perception-checking ability by developing complete perception-checking statements for each of the following situations. Be sure your statements include a description of the behavior, two...
-
Invite someone with whom you communicate frequently via mediated channels to rate your communicative competence. a. Explain the list of competencies in Section 2.3. b. Ask your evaluator to give you...
-
Recall recent situations in which you used each of the following evasive approaches: benevolent lying, equivocating, and hinting. Write an anonymous description of each situation on a separate sheet...
-
Taguchi argues that being within specification limits is not enough to be competitive in todays global economy. Do you agree? Why?
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
Given reg [3:0] A = 4'b1101; reg [2:0] B = 3'b111; reg signed [3:0] C = 4'b1101; reg signed [2:0] D = 3'b111; reg signed [7:0] S; Evaluate i. S = A + B; ii. S = A + B + 0; iii. S = C + D; iv. S = C +...
-
Given integer A = 8'shA5; integer B = 8'shB6; integer C = 8'hA5; integer D = 8'hB6; reg signed [31:0] S; Evaluate i. S = A + B; ii. S = A + B + 0; iii. S = C + D; iv. S = C + D + 0; v. S = A + D; vi....
-
A 4-bit magnitude comparator chip (e.g., 74LS85) compares two 4-bit numbers A and B and produces outputs to indicate whether A < B, A = B, or A > B. There are three output signals to indicate each of...
-
7. Let {E} be a monotone sequence of sets, that is, Ek C Ek+1 for k = 1, 2,... or Ek+1 C Ek for k = 1, 2,... Prove that, in either situation, we have lim inf Ek = lim sup Ek. 0043 k
-
Let (et) be IID(0,0) and define Xt = g(et, et1, . . ., etq), where g(.,...,.) is some real valued function. Show that Xt is strictly stationary.
-
A company rated AA issues a 3-year bond. The risk-free rate for the 3-year maturity is 3%. The lines of credit are summarized in the following table: Rating 1 year 5 years 10 20 years years AAA 12 14...

Study smarter with the SolutionInn App


