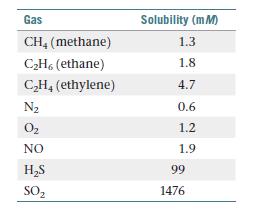
The following table presents the solubilities of several gases in water at 25 C under a total
Question:
The following table presents the solubilities of several gases in water at 25 °C under a total pressure of gas and water vapor of 1 atm.
(a) What volume of CH4(g) under standard conditions of temperature and pressure is contained in 4.0 L of a saturated solution at 25 °C?
(b) The solubilities (in water) of the hydrocarbons are as follows: methane 6 ethane 6 ethylene. Is this because ethylene is the most polar molecule?
(c) What intermolecular interactions can these hydrocarbons have with water?
(d) Draw the Lewis dot structures for the three hydrocarbons. Which of these hydrocarbons possess p bonds? Based on their solubilities, would you say p bonds are more or less polarizable than s bonds?
(e) Explain why NO is more soluble in water than either N2 or O2.
(f) H2 S is more water-soluble than almost all the other gases in table. What intermolecular forces is H2S likely to have with water?
(g) SO2 is by far the most water-soluble gas in table. What intermolecular forces is SO2 likely to have with water?
Step by Step Answer:

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus





