Estimate the partial molar enthalpy of sulfuric acid at 140F at the following two compositions using Figure
Question:
Estimate the partial molar enthalpy of sulfuric acid at 140°F at the following two compositions using Figure P9-22.
A. 30% by wt sulfuric acid
B. 80% by wt sulfuric acid
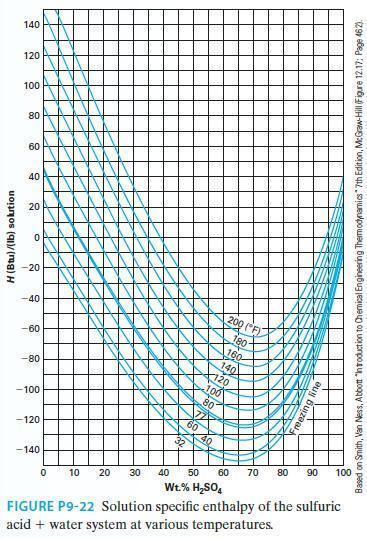
Transcribed Image Text:
H (Btu)/(lb) solution 140 120 100 80 60 40 20 O -20 -40 -60 -80 -100 -120 -140 } 200 (F) 180- 190 K But Bu 0 10 20 30 40 50 60 70 80 Wt.% H₂SO4 FIGURE P9-22 Solution specific enthalpy of the sulfuric acid + water system at various temperatures. 90 100 Based on Smith, Van Ness, Abbott "introduction to Chemical Engineering Thermodynamics" 7th Edition, McGraw-Hill (Figure 12.17: Page 462).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Using tabulated experimental data from the literature for either the excess molar volume or excess molar enthalpy of a system of your choice, provide the following information. A. A plot of the...
-
The molar enthalpy of a mixture of hydrogen fluoride (1) and water (2) at 20C is given as (Tyner, 1949) H = -1850 - 28240x, +26800x where II[=] J/mol Calculate the partial molar enthalpy of water in...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
In Problems 1130, solve each equation by factoring. x 2 - 9x = 0
-
Dicks Sporting Goods is a chain of full-line sporting goods retail stores offering a broad assortment of brand name sporting goods equipment, apparel, and footwear. Dicks Sporting Goods had its...
-
Fox Enterprises is owned by Edmund Fox and has a January 31 fiscal year end. The company prepares adjusting entries on an annual basis. The following trial balance was prepared before adjustments:...
-
Water flows in a 2-m-diameter finished concrete pipe so that it is completely full and the pressure is constant all along the pipe. If the slope is \(S_{0}=0.005\), determine the flowrate by using...
-
Earnings as defined in SFAC No. 5 are consistent with the current operating performance concept of income. Comprehensive income is consistent with the all- inclusive concept of income. Required: a....
-
XYZ Inc. sells two products, A and B. The selling price, variable cost and contribution margin per unit of product are shown below: Product A Selling Price $ 24 B $ 28 Variable Cost $ 10 $ 24...
-
Calculate the van Laar parameters, L 12 and L 21 , for the benzene (1) + toluene (2) system at 25C through use of the van der Waals equation of state, What would be the value for the activity...
-
A mass of 500 lb m of 40 wt% sulfuric acid solution at 140F is diluted with 200 lb m of pure water at 100F. What is the concentration of the resulting solution? What is the heat effect (liberated or...
-
On January 1, 2020, Nichols Company issued for $1,085,800 its 20-year, 11% bonds that have a maturity value of $1,000,000 and pay interest semiannually on January 1 and July 1. Bond issue costs were...
-
a. As the plan is not a safe harbor plan, the general rule is met as the highest allocation rate for any HCE is greater than the lowest allocation rate for any NHCE. True False. Regardless of your...
-
The December 31, 2024 balance sheet of Cullumber Co. included the following items: 10% bonds payable due December 31, 2030 $5,006,000 Unamortized discount on bonds payable 420,504 On January 1, 2025,...
-
Consider 24 PRNS,U 1,U2,...,U4, and suppose that the average of those observationssU= 24-1 U/24-0.7.Use these PRNs to generate a single approximately Nor(0,1) random variate via our "desert island"...
-
VQA regulations set standards including: Question 19 options: Addition of sugar, vineyard yeilds, grape varieties. Grape varieties, brix, labelling, origin. Brix, de-acidification, blending...
-
Find the free cash flow Tesla Valuation Model assumptions based on anaylst reports Upside YOY Revenue Growth Downside YOY Revenue Growth Base Case YOY Revenue Growth Total Cost of Revenue (% of Rev)...
-
Let X represent a binomial random variable with n = 150 and p = 0.36. Find the following probabilities. a. P(X 50) b. P(X = 40) c. P(X > 60) d. P(X 55)
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
Starting with the fully protonated species, write the stepwise acid dissociation reactions of the amino acids glutamic acid and tyrosine. Be sure to remove the protons in the correct order. Which...
-
Draw the structure of the predominant form of pyridoxal-5-phosphate at pH 7.00.
-
What fraction of ethane-1,2-dithiol is in each form (H 2 A, HA + , A 2+ ) at pH 8.00? at pH 10.00?
-
Stuart Daniels estimates that he will need $ 2 2 , 0 0 0 to set up a small business in 7 years. ( a ) How much ( in $ ) must Stuart invest now at 8 % interest compounded quarterly to achieve his goal?
-
Rowe Tool and Die (RTD) produces metal fittings as a supplier to various manufacturing firms in the area. The following is the forecasted Income statement for the next quarter, which is the typical...
-
On January 1, 2024, Kroll Corporation paid $2,559,000 for 31 percent of the outstanding voting stock of Sharon, Incorporated, and appropriately applied the equity method for its investment. Any...

Study smarter with the SolutionInn App


