The balloon portion of a dirigible contains 5000 moles of helium. Initially, the helium is at T
Question:
The balloon portion of a dirigible contains 5000 moles of helium. Initially, the helium is at T = 15°C and P = 0.95 atm, and is 500 m off the ground (where the atmospheric pressure is also 0.95 atm). The dirigible flies south at a constant height and a slow velocity, and gradually warms to T = 25°C (though the pressure remains constant) (Figure 3-12). How much heat was added to the balloon during the process?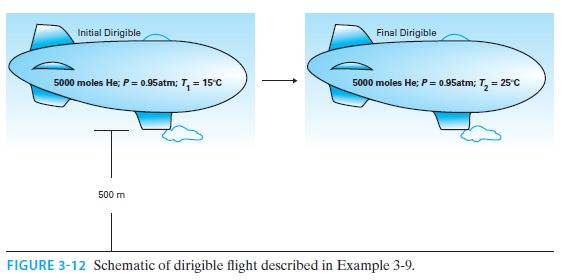
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:





