(A) What is E cell for the reaction in which Cl 2 (g) oxidizes Fe 2+ (aq)...
Question:
(A) What is E°cell for the reaction in which Cl2(g) oxidizes Fe2+(aq) to Fe3+(aq)?
![]()
(B) Use data from Table 19.1 to determine E°cell for the redox reaction in which Fe2+(aq) is oxidized to Fe3+(aq) by MnO4-(aq) in acidic solution.
Table 19.1
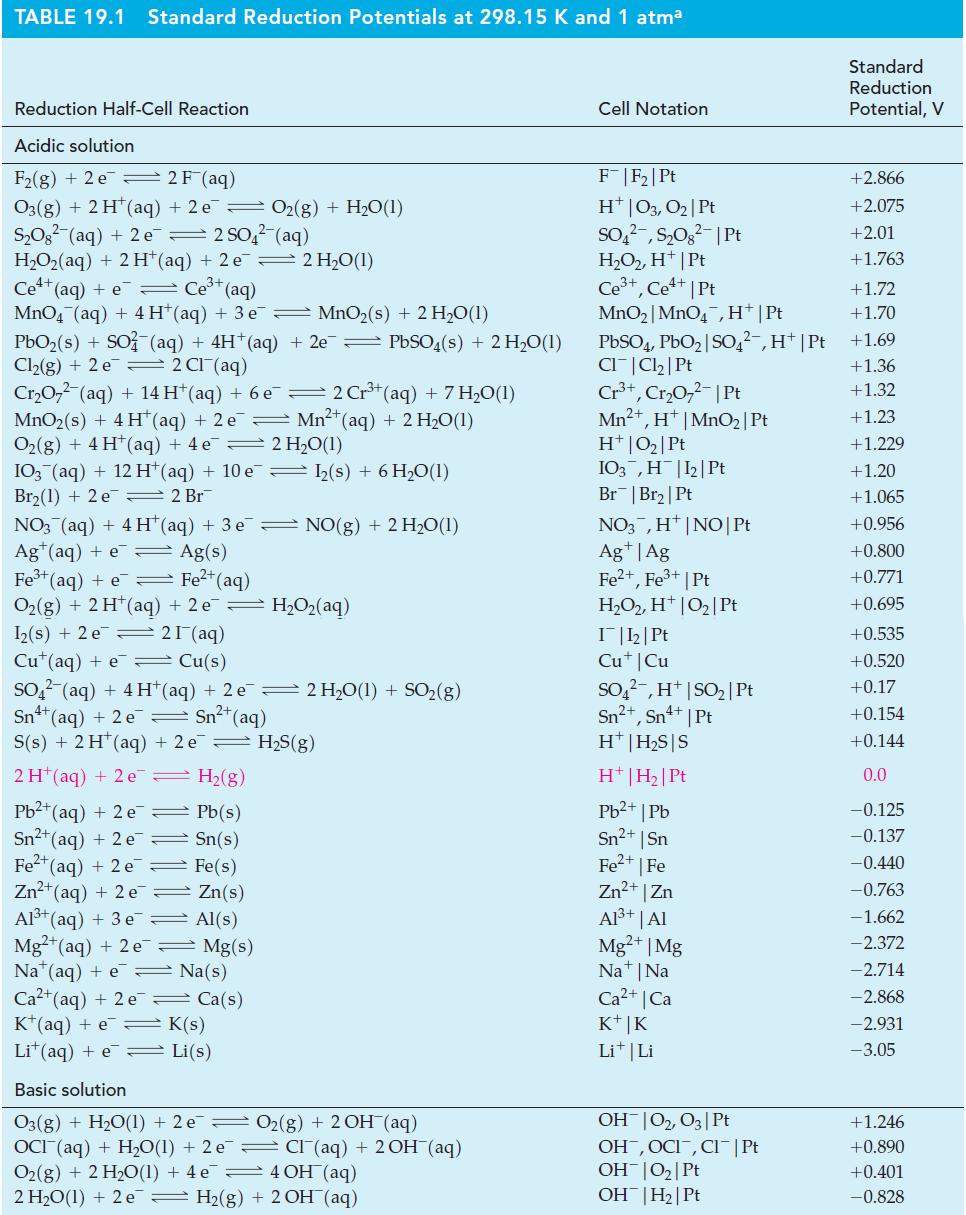
Transcribed Image Text:
3+ 2 Fe2+ (aq) + Cl₂(g) — 2 Fe³+ (aq) + 2 CI¯ (aq) Ecell ? =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The standard cell potential E for a given redox reaction can be calculated using the standar...View the full answer

Answered By

Payal Mittal
I specialize in finance and accounts.You can ask any question related to til undergradution.Organizational behaviour and HRM are my favourites for you can always relate to them and is an art with practical knowledge base.
4.90+
226+ Reviews
778+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Consider gravity loading only under complete lateral restraint in flat strata with properties given in Table 1.3. Vertical stress at the top of the geologic column given in Table 1.3 is 6.9 MPa....
-
In one type of Breathalyzer (alcohol meter), the quantity of ethanol in a sample is related to the amount of electric current produced by an ethanoloxygen fuel cell. Use data from Table 19.1 and...
-
(A) Use data from Table 19.1 to predict the probable products when Pt electrodes are used in the electrolysis of KI(aq). Table 19.1 (B) In the electrolysis of AgNO 3 (aq), what are the expected...
-
In Exercises 1114, graph each equation in a rectangular coordinate system. If two functions are indicated, graph both in the same system. Then use your graphs to identify each relations domain and...
-
Identify each of the following decision as most directly related to (a) Cash flow management (b) Choice of inventory system (c) Foreign merchandising transactions: 1. Determination of the amount of...
-
On September 25, 2010, a hurricane destroyed the work in process inventory of Biloxi Corporation. At that time, the company was in the process of manufacturing two custom jobs (B325 and Q428)....
-
On April 4, 2014, Athanasios Valsamis lost his appeal to get his money back from a friend to whom he had loaned \($700,000.\) As you will read, this case underscores the consequence of failing to...
-
The controller of Sonoma Housewares Inc. instructs you to prepare a monthly cash budget for the next three months. You are presented with the following budget information: The company expects to sell...
-
dentify the correctly revised sentence that adds interest and variety by beginning with an infinitive. Brooke congratulated me on securing investor financing for my startup company. Multiple choice...
-
A new battery system currently under study for possible use in electric vehicles is the zincchlorine battery. The overall reaction producing electricity in this cell is Zn(s) + Cl 2 (g) ZnCl 2 (aq)....
-
(A) The cell diagram for an electrochemical cell is written as Write the equations for the half-cell reactions that occur at the electrodes. Balance the overall cell reaction. (B) The cell diagram...
-
The actual selling expenses incurred in March 2012 by Vincent Company are as follows: Instructions (a) Prepare a flexible budget performance report for March using the budget data in E11-21, assuming...
-
(i) Define and contrast terms ideal solution, real solution, and regular solution, each using a single, complete sentence. (ii) For a binary system comprising components A and B. draw a plot of free...
-
Use a Base Year to Calculate Index Numbers for the Cost of a Basket of Goods and Services Please provided a detailed explanation so I can use it as a guide in my studies. Question The table below...
-
Explain how the shape of the expected utility function describes the consumers attitude to risk
-
Describe the mechanisms underlying primary and secondary growth in arboreal dicotyledonous plants, elucidating the distinctive physiological and anatomical processes involved in each phase.
-
Visit the Hunt Library and/or the Internet to find and review articles about capturing initiatives of MRO organization which is Delta Air Lines (Delta TechOps). Then, prepare a narrated presentation...
-
A marketing research firm wishes to estimate the proportion of adults who are planning to buy a new car in the next 6 months. A simple random sample of 100 adults led to 22 who were planning to buy a...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
(a) What is a lease agreement? (b) What are the two most common types of leases? (c) Distinguish between the two types of leases.
-
In general, what are the requirements for the financial statement presentation of long-term liabilities?
-
Laura Hiatt is discussing the advantages of the effective interest method of bond amortization with her accounting staff. What do you think Laura is saying?
-
QUESTION 3: 55 0.29 m ms i) Draw a free body diagram (FBD) of the sleeve. Be sure to label all forces, as well as your axes. ii) Find the magnitude of the ring's normal force on the sleeve as a...
-
A box with mass 1.68 kg is being pulled across a rough surface with a coefficient of kinetic friction k = 0.329. The pulling force has a magnitude of 14.4 N and is directed at an angle 24.8 degrees...
-
A parallel plate capacitor at 25 C comprises a slab of dielectric of area 104 m and a thickness of 2 mm. Assuming that the relative permittivity is 1200, the temperature coefficient of permittivity...

Study smarter with the SolutionInn App


