Complete and balance equations for these reactions. (a) LiH(s) + HO(1) (b) C(s) + HO(g) (c) NO(g)
Question:
Complete and balance equations for these reactions.
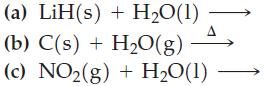
Transcribed Image Text:
(a) LiH(s) + H₂O(1) (b) C(s) + H₂O(g) (c) NO₂(g) + H₂O(1) A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a LiH HO ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation, acidbase, or gas-forming reaction. Show states for the products (s, , g, aq), give...
-
Manganese may play an important role in chemical cycles in the oceans. Two reactions involving manganese (in acid solution) are the reduction of nitrate ions (to NO) with Mn 2+ ions and the oxidation...
-
Complete and balance the following nuclear equations by supplying the missing particle: 98 53 59 0
-
Based on your data and observations, what are some of the differences between normal cells and cancer cells?
-
Which inventory cost flow alternative results in paying the least amount of taxes when prices are rising?
-
All else equal, which bonds price is more affected by a change in interest rates, a bond with a large coupon or a small coupon? Why?
-
The agreement contained a liquidated damages clause that held, Should Employee voluntarily terminate or the Employer terminates his/her employment within one year of executing this Agreement,...
-
Beka Company owns equipment that cost $50,000 when purchased on January 1, 2007. It has been depreciated using the straight-line method based on estimated salvage value of $5,000 and an estimated...
-
3. Consider a two period problem where a consumer has preferences over consumption in the two periods given by: log c + Blog c'. She has no initial assets and has income y in the first period y' in...
-
Give a practical laboratory method that you might use to produce small quantities of the following gases and comment on any difficulties that might arise: (a) O 2 ; (b) NO; (c) H 2 ; (d) NH 3 ; (e)...
-
Write a plausible chemical equation to represent the reaction of (a) Cl 2 (g) with cold NaOH(aq); (b) NaI(s) with hot H 2 SO 4 (concd aq); (c) Cl 2 (g) with KI 3 (aq); (d) NaBr(s) with hot H 3 PO 4...
-
When the Financial Accounting Standards Board issues new standards, the required implementation date is usually 12 months or more from the date of issuance, with early implementation encouraged....
-
Assume that we have an input signal x consisting of the sum of sine waves of 1.75 kHz and 2 kHz. We are sampling x at a rate of 3 kHz. Will we be able to reconstruct the original signal after...
-
Plant AG is considering making an offer to purchase Palmer AG. Plants vice president of finance has collected the following information: Plant also knows that securities analysts expect the earnings...
-
I have lived in four countries: South Africa, where I grew up, the US, where I was a teenage exchange student, Greece, where I learnt how to be a journalist, and the UK or, more specifically, London,...
-
Model your daily schedule with a timed automaton. Hours are reflected by a variable h, days by a variable d. d = 1 means Monday, d = 7 means Sunday. On a weekend (d = 6 or d = 7), you leave the...
-
Suppose Agile Corporation of the United States acquires Mobile Plc of the United Kingdom. The value of Agile stock on the Nasdaq in the United States is $3 billion. Mobile sells on the London Stock...
-
The Internal Revenue Service provides information on a variety of tax issues in its publication series. These publications can be found on the IRS Web site (www.irs.gov/formspubs/index.html). Go to...
-
Sportique Boutique reported the following financial data for 2012 and 2011. Instructions(a) Calculate the current ratio for Sportique Boutique for 2012 and 2011.(b) Suppose that at the end of 2012,...
-
The ledger of Zeta Corporation at December 31, 2012, after the books have been closed, contains the following stockholders equity accounts. Preferred Stock (10,000 shares issued)...
-
Melvina Corporation has been authorized to issue 20,000 shares of $100 par value, 7%, noncumulative preferred stock and 1,000,000 shares of no-par common stock . The corporation assigned a $5 stated...
-
On January 1, 2012, Neville Inc. had these stockholders equity balances. Common Stock , $1 par (2,000,000 shares authorized, 600,000 shares issued and outstanding) ..............$ 600,000 Paid-in...
-
7. In terms of financial planning, should CO2 emissions be considered? Why (not)?
-
A firm operating in stage I of the life cycle will have a dividend policy of a)stock dividends or low cash dividends b)stock dividends or moderate to low cash dividends c) moderate to high cash...
-
BHP is currently trading at $43/share and has announced a $0.95 dividend in 2months' time. European call options for BHP expiring in 5 months a with strike price of $45 were trading at $3. The...

Study smarter with the SolutionInn App


