The standard enthalpy of fermentation of glucose to ethanol is Use the standard enthalpy of combustion for
Question:
The standard enthalpy of fermentation of glucose to ethanol is
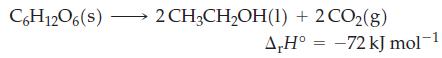
Use the standard enthalpy of combustion for glucose to calculate the enthalpy of combustion for ethanol.
Transcribed Image Text:
C6H12O6(s)- 2 CH3CH₂OH(1) + 2 CO₂(g) A,H° -72 kJ mol-1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The enthalpy change for a chemical reaction is given by the difference in the standard enthal...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Ethanol (C2H5OH) has been proposed as an alternative fuel. Calculate the standard enthalpy of combustion per gram of liquid ethanol.
-
The standard enthalpy of combustion of solid urea (CO (NH2)2) is -632 kl mol-1 at 298 K and its standard molar entropy is 104.60 J K-1 mol-1, Calculate the standard Gibbs energy of formation of urea...
-
The standard enthalpy of combustion of ethene gas [C2H4(g)] is 1411.1 kJ/ mol at 298 K. Given the following enthalpies of formation, calculate Hof for C2H4(g). CO2(g) 393.5 kJ/ mol H2O(l) 285.8 kJ/...
-
The following e-mail message contains numerous errors related to what you've learned about planning and writing business messages. Using the information it contains, write a more effective version....
-
Shown here are annual financial data at December 31, 2009, taken from two different companies. Required 1. Compute the cost of goods sold section of the income statement at December 31, 2009, for...
-
Based on the change in the assumed future compensation growth rate presented in Exhibit 1, which of the following pension cost components is aff ected? A . Service cost B . Remeasurement C . Net...
-
Mr. Tam is the front man for the Asian American dance-rock band The Slants. Mr. Tam named his band The Slants to reclaim and take ownership of Asian stereotypes. The band draws inspiration for its...
-
The graph in Figure represents various flows that can occur through a sewage treatment plant with the numbers on the arcs representing the maximum flow (in tons of sewage per hour) that can be...
-
a.) The following questions refer to the optical cavity in the diagram below. It is excited by a variable frequency source and the detected intensity is displayed on an Optical Spectrum Analyzer....
-
On May 2, 1987, Hannah acquired residential real estate for $450,000. Of the cost, $100,000 was allocated to the land and $350,000 to the building. On August 20, 2015, the building, which then had an...
-
Use the information given here, data from Appendix D, and equation (7.22) to calculate the standard enthalpy of formation per mole of ZnS(s). Eq.7.22 2 ZnS(s) + 3O(g) 2 ZnO(s) + 2 SO(g) A,H -878.2 kJ...
-
Use standard enthalpies of formation from Tables 7.2 and 7.3 and equation (7.22) to determine the standard enthalpy of reaction in the following reaction. Tables 7.2 Tables 7.3 Eq. 7.22 NH4+ (aq) +...
-
A state government wants to provide incentives for single parents to enter the labor market and become employed. It is considering a policy of paying single parents of children under age 18 $20 per...
-
What is the difference between financial projections and income statements? Income statements are informed forecasts of the future; financial projections are the historical data for your business...
-
Here's some info for a company: Cash $10,000 Accounts Payable $150,000 Inventory $100,000 Accounts Receivable $ 90,000 Short term Marketable Securities $50,000 What is the company's quick ratio?...
-
1. If our Economic System was Plain or Simple would it be prosperous? Explain Give example and Situation 2. If the only thing that drives the Economy of a country is the House of Commerce and the...
-
Apply appropriate technical analysis to explain why Austenitic 18/8 stainless steel is highly susceptible to weld decay when it is welded. (7 marks)
-
Explain why peripherals are not connected directly to the system bus? 2 . Compare between the different Input - Output control techniques: programmed I / O , in terrupt, DMA, and Channel IO . 3 ....
-
David Patel was at home when he received a call from the fire department telling him his store had burned. His business was a total loss. The insurance company asked him to prove his inventory loss....
-
What are the six activities involved in the physical supply/distribution system?
-
What is the indirect effect of a change in accounting principle? Briefly describe the reporting of the indirect effects of a change in accounting principle.
-
Define a change in estimate and provide an illustration. When is a change in accounting estimate effected by a change in accounting principle?
-
Lenexa State Bank has followed the practice of capitalizing certain marketing costs and amortizing these costs over their expected life. In the current year, the bank determined that the future...
-
A fisherman's scale stretches 3.1 cm when a 2.7 kg fish hangs from it. Express your answer to two significant figures and include the appropriate units. k = Value Submit Request Answer 4 Part B Units...
-
Mass m 1 = 1 4 1 . 2 kg is located at the origin of some coordinate system. A distance 1 5 . 3 m away is mass m 2 = 1 7 . 7 kg . How far from mass 1 should mass m 3 = 7 8 kg be placed if the...
-
Consider the following information for three stocks, Stocks X, Y, and Z. The returns on the three stocks are positively correlated, but they are not perfectly correlated. (That is, each of the...

Study smarter with the SolutionInn App


