Use bond energies from Table 10.3 to estimate r H for the following reaction. Table 10.3
Question:
Use bond energies from Table 10.3 to estimate ΔrH for the following reaction.
![]()
Table 10.3
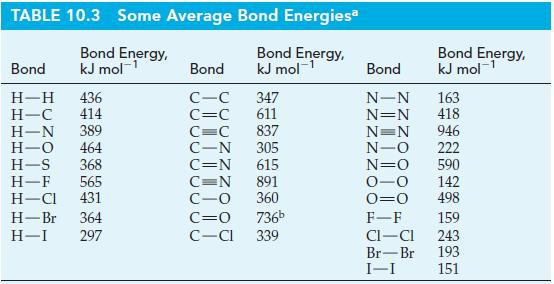
Transcribed Image Text:
C₂H2(g) + H2(g) C₂H4(g) A₁H = ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To estimate the enthalpy change AH for the given reaction C2H2g H2g C2H49 Well use bond energies fro...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Estimate the value of Ho for the following reaction from bond energies (Table 9.5). H2(g) + Cl2(g) 2HCl(g). Is the reaction exothermic or endothermic? Note that the reaction involves the breaking of...
-
Hydrazine (N 2 H 4 ) is used as a fuel in liquid-fueled rockets. When hydrazine reacts with oxygen gas, nitrogen gas and water vapor are produced. Write a balanced equation and use bond energies from...
-
Resonance energy is the difference in energy between a real moleculea resonance hybridand its most important contributing structure. To determine the resonance energy for benzene, we can determine an...
-
Describe a procedure for solving a first-degree inequality.
-
Pargo Corporation produces industrial robots for high-precision manufacturing. The following information is given for Pargo Corporation. The company has a desired ROI of 20%. It has invested assets...
-
a. Given the following graphs, calculate the total fixed costs, variable costs per unit, and sales price for Firm A. Firm Bs fixed costs are $120,000, its variable costs per unit are $4, and its...
-
On June 8, 2017, Eugene Weiner made a post on Isaac Aflalos Facebook page. The post read, Yurim and Isaac took advantage of a old 94plus sick man elder abuse [sic]. Alflalo took umbrage to the post...
-
Anglers Dream Company supplies flies and fishing gear to sporting goods stores and outfitters throughout the western United States. The accounts receivable clerk for Anglers Dream prepared the...
-
Recent events have once again shaken and stirred the memory of the nation. Almost immediately after the Civil War, a myth arose painting the conflict with a brush that attempted to blur the lines...
-
Equations (1) and (2) can be combined to yield the equation for the formation of CH 4 (g) from its elements. Use the preceding data and a bond energy of 436 kJ mol -1 for H 2 to estimate the CH bond...
-
One of the isomers of chloromethanol has the formula ClCH 2 OH. Sketch, by using the dash and wedge symbolism, this isomer of chloromethanol, and indicate the various bond angles.
-
An ordinary 12-oz beer or soda can holds about 25 in 3 . Find the dimensions of a can with minimum surface area. Measure a can and see how close its dimensions are to the results you found.
-
My Custom Shop, Inc., (MCS) bought an 18-year-old Ford Ranger truck with 178,000 miles at auction for about $1,300. MCS made several improvements to the vehicle and then sold it to Seth Kiewiz for...
-
How does negligence-based product liability differ from strict product liability?
-
Fine Textiles, Inc., sells cloth to Gail by showing her a sample that Fines salesperson says is the same as the goods. This statement is a. an express warranty. b. an implied warranty. c. a warranty...
-
Eagle Equipment sells motor vehicle parts to dealers. In response to a dealers order, Eagle ships a crate with a label that reads, Crate contains one 150-horsepower diesel engine. This statement is...
-
B&B Sales, Inc., sells drones. A B&B salesperson claims, This is the finest drone ever made. This statement is a. an express warranty. b. an implied warranty of merchantability. c. an implied...
-
Mariah Enterprises makes a variety of consumer electronic products. Its camera manufacturing plant is considering choosing between two different processes, named Alpha and Beta, which can be used to...
-
Modify the counter from Exercise 5.44 such that the counter will either increment by 4 or load a new 32-bit value, D, on each clock edge, depending on a control signal Load. When Load = 1, the...
-
Statistical quality control, airline operations. Jetrans Airlines operates daily round-trip flights on the LondonLos Angeles route using a fleet of three 747s: the Spirit of Atlanta, the Spirit of...
-
Compensation linked with profitability, waiting time, and quality measures. Mid-Atlantic Healthcare operates two medical groups, one in Philadelphia and one in Baltimore. The semi-annual bonus plan...
-
Waiting times, manufacturing lead times. The SRG Corporation uses an injection molding machine to make a plastic product, Z39. SRG makes products only after receiving firm orders from its customers....
-
Cray Computing needs a 8-month loan for $300,000. Its bank quotes a simple interest rate of 5% on the loan. What is the annual percentage rate (APR)? What is the effective annual rate (EAR)? What is...
-
Product AB has a suggested selling price of $27 per unit and a projected variable cost per unit of $15. Fixed costs are expected to increase by $197,040 per month. How many units must be sold each...
-
A project being tendered at $100,000 has material costs of $60,000 and labour costs of $20,000. What amount should be allowed in the estimate to cover the premium cost for a 50% labour and material...

Study smarter with the SolutionInn App


