Use data from Appendix D to determine values of rG for the following reactions at 25 C.
Question:
Use data from Appendix D to determine values of ΔrG° for the following reactions at 25 °C.
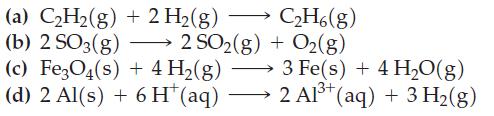
Transcribed Image Text:
→ (a) C₂H₂(g) + 2 H₂(g) (b) 2 SO3(g) (c) Fe3O4(s) + 4 H₂(g) (d) 2 Al(s) + 6 H* (aq) 2 SO2(g) C2₂H6(g) + O2(g) 3 Fe(s) + 4 H₂O(g) 2 Al³+ (aq) + 3 H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
c Fe3O4s 4H2g 3Fes 4HOg AG Ker d 2 Als 6H aq 2A1 aq 3H2g Kae Now we can use the standard Gibbs free ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In Example 13-3, we dealt with vap H and vap S for water at 100 C. (a) Use data from Appendix D to determine values for these two quantities at 25 C. (b) From your knowledge of the structure of...
-
Use data from Appendix D to determine values at 298 K of and K for the following reactions. (The equations are not balanced.) (a) HCl(g) + O(g) (b) FeO3(s) + H(g) (c) Ag(aq) + SO4 (aq) HO(g) + Cl(g)...
-
Resonance energy is the difference in energy between a real moleculea resonance hybridand its most important contributing structure. To determine the resonance energy for benzene, we can determine an...
-
Find the minimum and maximum values of the function subject to the given constraint. f (x, y) = x + y, x + y4 = 1
-
What are the three approaches for determining transfer prices?
-
Write a formula that can be used to value any stock, regardless of its dividend pattern.
-
Cat Auto Tech. Corp. purchased 10,000 gift certificates from DeJesus. Cat Auto Tech. Corp., an Amoco gasoline station operator, contracted with DeJesus to make 10,000 gift certificates of various...
-
Assume that Denis Savard Inc. has the following accounts at the end of the current year. 1. Common Stock. 2. Discount on Bonds Payable. 3. Treasury Stock (at cost). 4. Notes Payable, short-term. 5....
-
W (K$) 14 13 Rich 12 11 10 9 8 7 6 5 Poor W (K$) 14 13 12 11 10 9 8 7 6 5 4 16 18 20 22 24 26 28 17 19 21 L 20 22 24 26 23 25 27 (M) 21 23 25 27 29 31 28 30 32 34 36 L 33 35 37 (M) 1. There are two...
-
Explain why (a) Some exothermic reactions do not occur spontaneously, and (b) Some reactions in which the entropy of the system increases do not occur spontaneously.
-
We will see that, for the formation of an ideal solution of liquid components, H = 0. What would you expect for the values of S and G? (Is each value positive, negative, or zero?)
-
In Problems 4752, find functions f and g so that f g = H. H(x): x + 1
-
Starbucks has said its goal is to expand delivery to one-quarter of its US stores and increase earnings per share by 10 percent. How will the organizing function contribute to the accomplishment of...
-
Headquartered in Boulogne-Billancourt, France, the automotive manufacturer Renault was established 120 years ago in 1899, and Japanese automotive manufacturer, Nissan, was founded in 1933,...
-
If Starbucks wanted to offer compressed workweeks, flextime, or job sharing, what would they need to consider? What are advantages and disadvantages of offering these employment options at Starbucks?
-
What possible problems could Starbucks encounter with their matrix structure? What could they do to try to minimize these problems?
-
If Starbucks wanted to implement multiperson comparisons or 360-degree appraisals as methods of evaluating employee performance, what possible issues (both positive and negative) might arise? How...
-
You have recently been promoted to be director of advertising for the Timkin Tool Company. In your first meeting with Mr. Timkin, he says, Advertising is a waste! Weve been advertising for six months...
-
The 2017 financial statements of the U.S. government are available at: https://www.fiscal.treasury.gov/fsreports/rpt/finrep/fr/fr_index.htm Use these to answer the following questions: a. Statement...
-
Target costs, effect of product-design changes on product costs. Medical Instruments uses a manufacturing costing system with one direct-cost category (direct materials) and three indirect-cost...
-
Cost-plus target return on investment pricing. John Beck is the managing partner of a business that has just finished building a 60-room motel. Beck anticipates that he will rent these rooms for...
-
Cost-plus, target pricing, working backward (S. Sridhar, adapted) Waterbury, Inc., manufactures and sells RF17, a specialty raft used for whitewater rafting. In 2009, it reported the following: 1....
-
On March 18, when the posted prime rate was 2%, an investment of $2,000 was placed into an account earning prime + 1.5%. Effective May 2, the prime rate rose by 1%. If the money was withdrawn on...
-
Required information [The following information applies to the questions displayed below.] The first production department of Stone Incorporated reports the following for April. Direct Materials...
-
AudioFile Products Ltd. is a retailer of sound systems. The company is planning its cash needs for the month of January 2024. In the past, AudioFile has had to borrow money during the post-Christmas...
Operations Management In Healthcare Strategy And Practice 2nd Edition - ISBN: 0826147712 - Free Book

Study smarter with the SolutionInn App


