Use data from Figure 9-1 and equation (9.1) to estimate the density of the recently discovered element
Question:
Use data from Figure 9-1 and equation (9.1) to estimate the density of the recently discovered element 114.
Figure 9-1
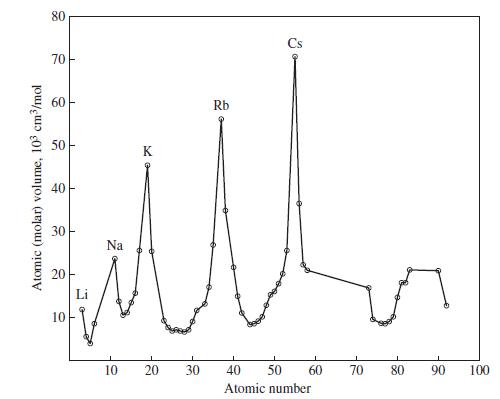
Eq. 9.1
![]()
Transcribed Image Text:
Atomic (molar) volume, 103 cm³/mol 80 70 60 50 40 30 20 10 T Na 10 K 20 30 Rb Cs 40 50 Atomic number 60 70 80 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To estimate the density of element 114 using the data from Figure 91 and equation 91 ...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Buoyancy correction factor: mtrue/mread = 1.000 3244] [Interpolated density of water at 23.3 C: 0.997 468 9 g/mL] [HCl concentration at 20 C: 0.102343 M] Reagents: The molar mass and density of...
-
Estimate the density of the water 6.0 km deep in the sea. (Se Table9-1 and Section 9-5 regarding bulk modulus.) By what fraction does it differ from the density at the surface?
-
Use the Arrhenius equation (Section 2-1) to estimate the ratio of the rate constants k for the reactions of a C-H bond in methane with a chlorine atom and with a bromine atom at 25C. Assume that the...
-
(a) By what percentage does your rest mass increase when you climb 30 m to the top of a ten-story building? Are you aware of this increase? Explain. (b) By how many grams does the mass of a 120-g...
-
Refer to the data in Exercise 6-31. Compute the predetermined overhead rate assuming that Tiger Furnishings uses machine-hours to allocate overhead costs.
-
An air-standard Ericsson cycle has an ideal regenerator. Heat is supplied at 1000C and heat is rejected at 20C. Pressure at the beginning of the isothermal compression process is 70...
-
Should the requirements of the UCC be subject to the application of reliance theories? Go back and review the facts in Case 21-3 about the coal contract. Should silence followed by contract execution...
-
Toxaway Company is a merchandiser that segments its business into two divisionsCommercial and Residential. The companys accounting intern was asked to prepare segmented income statements that the...
-
Define the general - purpose financial statements and points out the limitation of such statements.
-
Your client, Leona Ledford, was personally served with a summons and complaint on October 23 in the case of Masters v Ledford Her answer is due in 30 days. You will mail the answer to the court. What...
-
Refer only to the periodic table on the inside front cover, and determine which is the largest atom: Sc, Ba, or Se.
-
When the ionization energies of a series of isoelectronic atoms and ions are compared, an interesting relationship is observed for some of them. In particular, if the square root of the ionization...
-
Create an application, using the following names for the solution and project, respectively: Kerry Cable Solution and Kerry Cable Project. Save the application in the VB2015\Chap05 folder. Kerry...
-
You Healthy living Health Foods Inc. is authorized to issue 4,250,000 common shares. Requirement Show the computation of Healthy Living's total shareholders' equity at December 31, 2020 . Present a...
-
3. Alpha Investors On July 1, 1985, Jim Roberts felt certain the stock market was in for a correction. The S&P 500 Index had risen 14% since the first of the year, and most analysts were predicting...
-
Assume that you have just been hired as business manager STS. The Cost of Debt of STS with Different Capital Structures is shown in the table below Percent Financed with Debt (wd) Cost of Debt (rd)...
-
Prairie Corporation holds cash of $10,000 and owes $29,000 on accounts payable. Prairie has accounts receivable of $47,000, inventory of $18,000, and land that cost $40,000. How much are Prairie's...
-
Select Silver contract owner, Edward, is age 54. Renew to a guaranteed period of one, three , five, or seven years . The withdrawal charges are now at zero , so the client may withdraw the funds. The...
-
Takumi Computers currently purchases for $32 each keyboard it uses in the 50,000 computers it makes and sells annually. Each computer uses one keyboard. The company has idle capacity and is...
-
An 8.0 kg crate is pulled 5.0 m up a 30 incline by a rope angled 18 above the incline. The tension in the rope is 120 N, and the crates coefficient of kinetic friction on the incline is 0.25. a. How...
-
How does conformance quality differ from design quality? Explain.
-
Name two items classified as prevention costs.
-
Distinguish between internal failure costs and external failure costs.
-
Following is a trial balance of City of Jeddah's General Fund at December 3 1 , 2 0 2 2 . Prepare: ( a ) a statement of revenues, expenditures, and changes in fund balance for the year ended...
-
Draw the truth table for the output G ( Inputs X , Y , Z , W ) . 3444 44 2-494 DD 50 D.P
-
Auditing employee compensation and benefits is crucial for ensuring that organizations comply with applicable laws and regulations, accurately record expenses, and maintain internal controls over...

Study smarter with the SolutionInn App


