Use Hesss law and the following data CH4(g) + 2O2(g) CH4(g) + CO(g) CH(g) +
Question:
Use Hess’s law and the following data
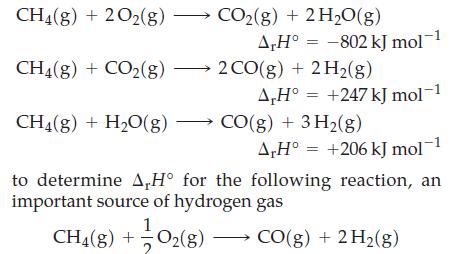
Transcribed Image Text:
CH4(g) + 2O2(g) → CH4(g) + CO₂(g) — CH₂(g) + H₂O(g) CO₂(g) + 2H₂O(g) A,H° -802 kJ mol 2 CO(g) + 2H₂(g) A,H° = +247 kJ mol-1 CO(g) + 3H₂(g) A,H° +206 kJ mol−1 to determine A,H° for the following reaction, an important source of hydrogen gas CH4(g) + O₂(g) — CO(g) + 2 H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
To use Hesss law to determine the enthalpy change for the reaction CH4g 12O2g COg 2H2g We can use th...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The following is the monthly payroll of Johnson Company, owned by Dan Johnson. Employees are paid on the last day of each month. Johnson Company is located at 2 Square Street, Marblehead,...
-
John Andrews, the accountant for Johnson Company, must complete Form 941 for the first quarter of the current year. John gathered the needed data as presented in Problem 8A-2. Suddenly called away to...
-
Gebruik Hess se Wet om die reaksiewarmte vir die proses te bepaal. Use Hess's Law to determine the heat of reaction for the process. CH4(g) + 6HCl(g) 2CHCl3(g) + 4H(g) i. 2C(s) + 2H(g) CH4(g) | AH =...
-
Which of the following is most nearly the mass of the Earth? (The radius of the Earth is about 6.4 106 m) A. 6 x 1024 kg B. 6 x 107 kg C. 6 x 103 kg D. 6 x 1033 kg E. 6 x 1036 kg
-
Many fast-food restaurants compete on lean business concepts. Match each of the following activities at a fast-food restaurant with the lean business concept it strives to achieve. Some activities...
-
Th e plan for which the amount of SKIs fi nancial obligation is defi ned in the current period with no obligation for future retirement benefi ts is: A . Plan A. B . Plan B. C . Plan C.
-
Shiloh supplies equipment to the automotive and commercial vehicle markets and other industrial customers. It specializes in materials and designs that reduce vehicle weight and increase fuel...
-
Wonderful Not only did our salespeople do a good job in meeting the sales budget this year, but our production people did a good job in controlling costs as well, said Kim Clark, president of Martell...
-
Who developed the relational model, when, and why?
-
Janice Morgan, age 24, is single and has no dependents. She is a freelance writer. In January 2021, Janice opened her own office located at 2751 Waldham Road, Pleasant Hill, NM 88135. She called her...
-
The standard heats of combustion ( r H) of buta-1,3-diene, C 4 H 6 (g); butane, C 4 H 10 (g); and H 2 (g) are -2540.2, -2877.6, and -285.8 kJ mol-1, respectively. Use these data to calculate the heat...
-
CCl 4 , an important commercial solvent, is prepared by the reaction of Cl 2 (g) with a carbon compound. Determine r H for the reaction CS(1) + 3Cl(g) CCl4(1) + SCl2 (1) Use appropriate data from...
-
Repeat Problem 13 for the system shown in Figure 5.72, in which each pendulum is a uniform slender rod of mass \(m\) and length \(L\). The inputs are the torques \(\tau_{1}\) and \(\tau_{2}\) applied...
-
Samuel Jenkins made two investments; the first was 13 months ago and the second was two months ago. He just sold both investments and has a capital gain of $4,000 on each. If Samuel is single and has...
-
PT Partnership begins with 2 partners. Parrot contributes assets with a FMV of $50,000 and an adjusted basis of $10,000, and Troy contributes accounting services of $50,000. What income must Parrot...
-
Cavalier Bicycles uses carbon fiber to produce its high-performance road and mountain bikes. Cavalier anticipates it will need to purchase 45,000 pounds of carbon fiber in July 2025 to make enough...
-
Although, accountability in public administration is different from accountability in the private sector, yet, an increasing number of public services are being contracted to private sector...
-
How should StartupWeb account for payments under this contract, including cash payments and the issuance and exercise of the options? Pay particular attention to the amounts involved and the...
-
In chronological order, the inventory, purchases, and sales of a single product for a recent month are as follows. 1. Using the periodic inventory system, compute the cost of ending inventory , cost...
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
An entry to record Purchases and related Accounts Payable of $13,000 for merchandise purchased on December 23, 2011, was recorded in January 2012. This merchandise was not included in inventory at...
-
Equipment was purchased on January 2, 2010, for $24,000, but no portion of the cost has been charged to depreciation . The corporation wishes to use the straight-line method for these assets, which...
-
Where can authoritative iGAAP related to accounting changes be found?
-
What are the philosophical, ethical, and legal implications of defining and protecting species diversity, including debates surrounding the intrinsic value of species, the rights of non-human...
-
Explain how gross income of interest or royalty income shall be deemed to be Malaysian sourced. give explanation with example
-
If an investor buys shares in a no-load mutual fund for $39 and after eight years the shares appreciate to $47, what is (1) the percentage return and (2) the annual compound rate of return using time...

Study smarter with the SolutionInn App


