Question: Use the Nernst equation and data from Appendix D to calculate E cell for each of the following cells. (a) Mn(s) Mn+ (0.40 M)||Cr+ (0.35
Use the Nernst equation and data from Appendix D to calculate Ecell for each of the following cells.
, OH](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1700/8/1/3/79965605be75484e2.png)
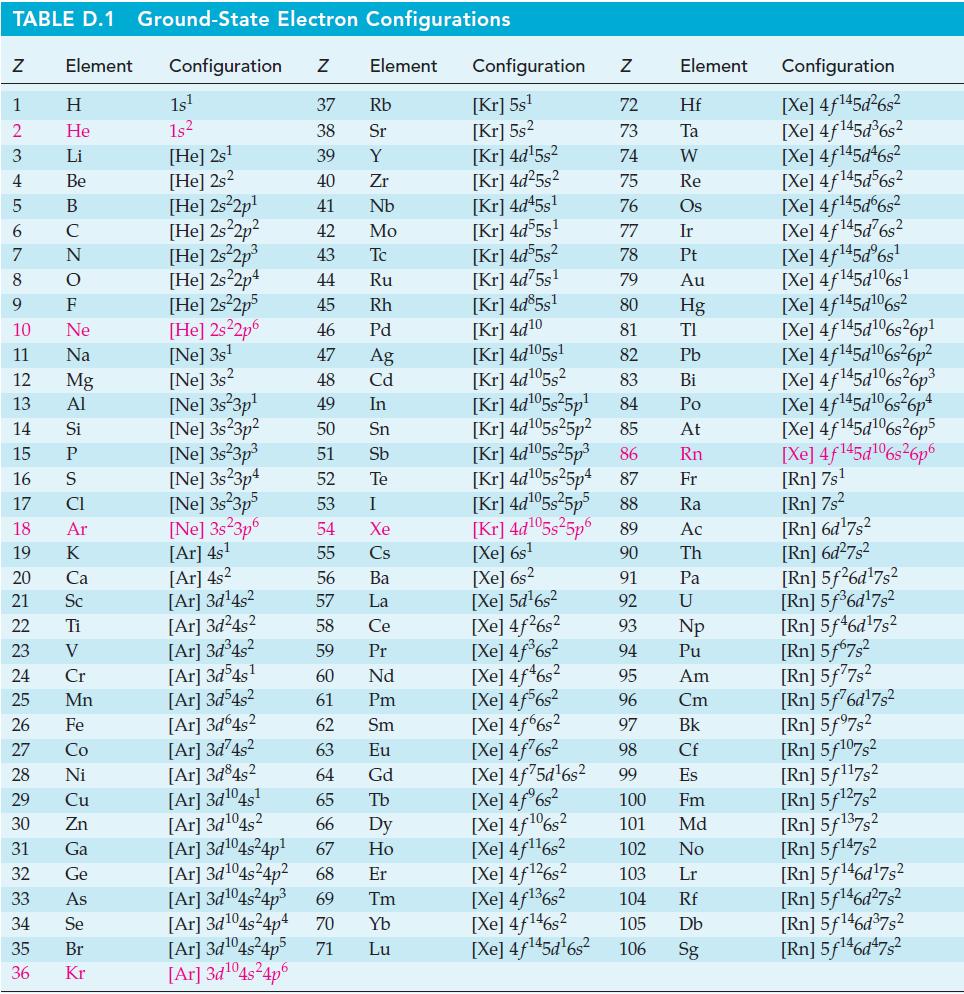
(a) Mn(s) Mn+ (0.40 M)||Cr+ (0.35 M), Cr+ (0.25 M) Pt(s) (b) Mg(s) Mg2+ (0.016 M)||[Al(OH)4](0.25 M), OH (0.042 M) Al(s)
Step by Step Solution
3.46 Rating (162 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


