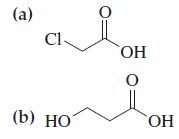
Write Lewis structures for the molecules represented by the following line-angle formulas. (a) C1- (b) HO OH
Question:
Write Lewis structures for the molecules represented by the following line-angle formulas.
Transcribed Image Text:
(a) C1- (b) HO OH 0 ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write Lewis structures for the molecules represented by the following line-angle formulas. (a) (b) Cl NH
-
Write Lewis structures for the molecules represented by the following molecular models. (a) (b)
-
Write Lewis structures for the molecules represented by the following molecular models. (a) (b)
-
A firm has initial fund of $800. The market rate of return is 12%. The firm has identified good investment opportunities to invest $640 at an average 25 per cent rate of return. How much dividend...
-
The balance sheets of Tully Corp. showed the following at December 31, 2011, and 2010: Required: a. If there have not been any purchases, sales, or other transactions affecting this machine account...
-
Single Audit. Quad-States Community Service Agency expended federal awards during the most recent fiscal year in the following amounts for the pro- grains shown: Additional information indicates that...
-
Use the technique discussed in Section9.4 to develop a GEE approach for zeroinflated Poisson model for count responses in longitudinal studies. Section9.4: 9.4 Marginal Models for Longitudinal Data...
-
Fallow Co. had the following transactions during the current period. Mar. 2 Issued 5,000 shares of $1 par value common stock to attorneys in payment of a bill for $38,000 for services provided in...
-
You have made a physical pendulum by swinging a rod of mass M = 0.73 kg and length L = 0.53 meters around its end. The mass of the rod is distributed uniformly along its length. We will assume that...
-
Identify the main group that the element X belongs to in each of the following Lewis structures. For the types of molecule shown, give an example that exists. (a) [:XX; :: 2- (c) |:0 x6: (b) :0- ::...
-
Write a plausible Lewis structure for C 3 O 2 , a substance known as carbon suboxide.
-
Ardmore Investments purchased Columbia Corporation shares on December 16 for $110,000. Ardmore plans on holding the securities a few months. Ardmore owns less than 2% of the outstanding shares of...
-
Borax, sodium tetraborate decahydrate, is an important mineral found in dry lakebeds in California. It is used to make soap and glass, and it is also used as a preservative. You can use the values of...
-
Suppose you have midterms in economics and astronomy tomorrow, and you only have four hours left to study. The accompanying table provides the combinations of time spent studying economics and...
-
The compound H 3 N BH 3 is an adduct of NH 3 and BH 3 . It is currently being investigated as a possible hydrogen storage material. (a) What is the hydrogen storage capacity (percentage by weight)...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Tungsten hexacarbonyl (4.18) contains six equivalent CO ligands. With reference to Table 4.3, suggest what you would...
-
What is the Unified Process (UP)?
-
Review problem-time value of money applications. Use the appropriate factors from Table 6-4 or Table 6-5 to answer the following questions. Required: a. Spencer Co.'s common stock is expected to have...
-
Feller Company purchased a site for a limestone quarry for $100,000 on January 2, 2019. It estimate that the quarry will yield 400,000 tons of limestone. It estimates that its retirement obligation...
-
This makes no sense at all, said Bill Sharp, president of Essex Company. We sold the same number of units this year as we did last year, yet our profits have more than doubled. Who made the goofthe...
-
These statements cant be right, said Ben Yoder, president of Rayco, Inc. Our sales in the second quarter were up by 25% over the first quarter, yet these income statements show a precipitous drop in...
-
Guochang Li was hired as chief executive officer (CEO) in late November by the board of directors of ContactGlobal, a company that produces an advanced global positioning system (GPS) device. The...
-
1. Possibly in a group discussion, build a scenario relevant for the motor vehicle industry in ten years time. How will people be using their cars? What will they expect in terms of size,...
-
Explain the difference between the predispositional and socialized theories of police behavior.
-
2. You are the investment manager of a well-diversified portfolio. Due to recent interest in the farming and mining industries, you are considering adding the stock of Caterpillar (CAT) or Deere (DE)...

Study smarter with the SolutionInn App


