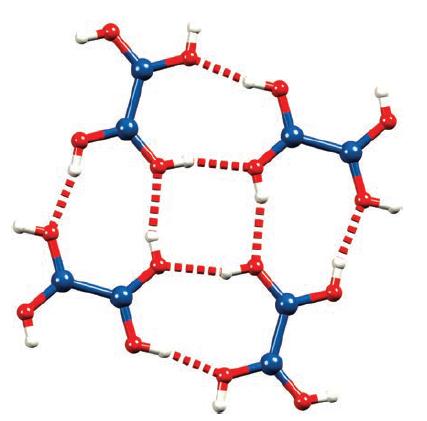
Figure 13.21 shows four hydrogen-bonded molecules of B 2 (OH) 4 . To what point group does
Question:
Figure 13.21 shows four hydrogen-bonded molecules of B2(OH)4. To what point group does a single molecule of B2(OH)4 belong?
Figure 13.21.
Transcribed Image Text:
.....
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
A single molecule of B2OH4 belongs to the point group D2h The point group of a molecule is the set o...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the molecule CH3Cl. (a) To what point group does the molecule belong? (b) How many normal modes of vibration does the molecule have? (c) What are the symmetries of the normal modes of...
-
(a) Draw a set of resonance structures for the hypothetical molecule PH 5 , ensuring that P obeys the octet rule in each structure. Assume a structure analogous to that of PF 5 . (b) To what point...
-
Determine what point group does PF 5 belong to, discuss how that point group is constructed, and discuss where do the linear and quadratic terms of that point group come from.
-
Kiev Corp. was incorporated on January 2, 2020, but was unable to begin manufacturing activities until July 1, 2020, because new factory facilities were not completed until that date. The Land and...
-
Trenton Fabrication Company purchased industrial tools costing $110,000, which fall in the 3-year property class under MACRS. Required: 1. Prepare a schedule of depreciation deductions assuming: a....
-
Work Problem 6-38 by using the internal rate of return (IRR) method. The MARR is 15% per year. Remember to think incrementally! Data from Problem 6-38: Two mutually exclusive alternatives for office...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
GroFast Company manufactures a high-quality fertilizer, which is used primarily by commercial vegetable growers. Two departments are involved in the production process. In the Mixing Department,...
-
This table lists the gross property tax distribution applicable to a local homeowner. Property Tax Distribution. Taxing Authority 2017 2018 Airport $45.88 $46.71 Citilink - PTC $63.69 $66.18 City...
-
Figure 13.11c shows the solid state structure of the [Al(BH 4 ) 4 ] ion, present in [Ph 3 MeP][Al(BH 4 ) 4 ]. In the light of these structural data, account for the following observations, recorded...
-
Write equations for the following processes, involved in the extraction of the elements from their ores: (a) The reduction of boron oxide by Mg; (b) The result of the addition of hot aqueous NaOH to...
-
Which one of the following is an example of a disjoint and partial specialization? a. HUMAN VEGETARIAN + NON-VEGETARIAN b. HUMAN BLONDE + BRUNETTE c. HUMAN LOVES FISH + LOVES MEAT d. HUMAN ...
-
A gig refers to a single project or task that an individual completes for pay, and individuals who complete gigs are often referred to as gig workers. Examples of gigs range from driving for Uber and...
-
Hamdi Ulukaya, founder and CEO of Chobani, LLC, left his family dairy business in Turkey to learn English in the United States. Soon after he arrived, he noticed that the strained yogurt popular in...
-
Three years ago, a large clothing retailer called La Ropa de Moda developed and implemented a new pay-for-performance program targeted at sales associates. The relatively new program is a sales...
-
Should an organization avoid hiring individuals with a criminal background? There is no federal law prohibiting discrimination against former inmates. Yet having blanket policies excluding those with...
-
Implementing merit pay for teachers has been a contentious issue, particularly in the United States. Supporters contend that merit pay motivates teachers to do a better job, leading to better...
-
Five advertising companies jointly buy a jet aircraft. They enter into an agreement whereby each party has the right to use the aircraft for its own purposes some days each year. The parties may...
-
Diamond Walker sells homemade knit scarves for $25 each at local craft shows. Her contribution margin ratio is 60%. Currently, the craft show entrance fees cost Diamond $1,500 per year. The craft...
-
Draw the resonance structures for CO 3 2 .
-
Develop an argument based on bond enthalpies for the importance of SiO bonds, in preference to SiSi or SiH bonds, in substances common in the Earths crust. How and why does the behaviour of silicon...
-
(a) Use a molecular orbital program or input and output from software supplied by your instructor to construct a molecular orbital energy-level diagram to correlate the MO (from the output) and AO...
-
DIET PROBLEM. Grant Winfield is a 71-year-old grandfather who likes to mix breakfast cereals together for taste and as a means of getting at least 50% of the recommended daily allowances (RDA) of...
-
John Johnson decided to leave his former job where he earned $12 per hour to go to a new job where he will earn $13 per hour. In the decision process, the former wage of $12 per hour would be...
-
In the balance sheet, the account, Premium on Bonds Payable, is ? Four thousand bonds with a face value of $1,000 each, are sold at 105. The entry to record the issuance is ? 81.Bond interest paid...

Study smarter with the SolutionInn App


