A mixture of ethanol and ethyl acetate is heated in a closed system at 100 kPa to
Question:
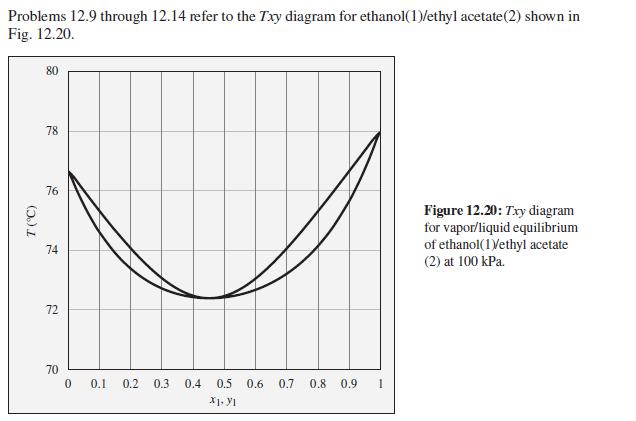
A mixture of ethanol and ethyl acetate is heated in a closed system at 100 kPa to a temperature of 74°C, and two phases are observed to be present. What are the possible compositions of the liquid and vapor phases?
Transcribed Image Text:
Problems 12.9 through 12.14 refer to the Txy diagram for ethanol(1)/ethyl acetate(2) shown in Fig. 12.20. 80 78 76 Figure 12.20: Txy diagram for vapor/liquid equilibrium of ethanol(1Vethyl acetate 74 (2) at 100 kPa. 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1 X1. YI (5.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
The pressure above a mixture of ethanol and ethyl acetate at 70C is measured to be 78 kPa. What are the possible compositions of the liquid and vapor phases? Problems 12.3 through 12.8 refer to the...
-
The pressure above a mixture of ethanol and ethyl acetate at 70C is measured to be 86 kPa. What are the possible compositions of the liquid and vapor phases? Problems 12.3 through 12.8 refer to the...
-
A chloroform and tetrahydrofuran mixture is heated in a closed system at 120 kPa to a temperature of 70C, and two phases are observed to be present. What are the possible compositions of the liquid...
-
Solve Utt = cUTT 0
-
Denise will receive $1,000 in two years. If the interest rate is 5 percent, what is the present value of this dollar amount?
-
Aivars Company reports the following variable costing income statement for its single product. This companys sales totaled 50,000 units, but its production was 80,000 units. It had no beginning...
-
A call option pays an amount \(V(S)=\frac{1}{1+\exp (S(T) K)}\) at time \(T\) for some predetermined price \(K\). Discuss what you would use for a control variate and generate a simulation to...
-
Various types of accounting changes can affect the auditor's report. a. Briefly describe the rationale for having accounting changes affect the auditor's report and the auditor's responsibility in...
-
Transaction: The company paid $500 on January 1 for January's rent. In the table below, show the effect of the transaction above to each of the categories below (Assets, Liabilities, Equity) and the...
-
Gauntlet Gyms Inc. owns athletic training facilities and camping grounds, which it leases to corporate clients, educational institutions, and other non-governmental organizations. The Xavier...
-
Air, even more than carbon dioxide, is inexpensive and nontoxic. Why is it not the gas of choice for making soda water and (cheap) champagne effervescent? Table 13.2 may provide useful data. Table...
-
A mixture of ethanol and ethyl acetate is heated in a closed system at 100 kPa to a temperature of 77C, and two phases are observed to be present. What are the possible compositions of the liquid and...
-
Determine the focal length of a planar-concave lens (n l = 1.5) having a radius of curvature of 10 cm. What is its power in diopters?
-
Guthrie plc has current liabilities of 340,000, a quick ratio of 1.8, inventory turnover of 4.2 and a current ratio of 3.3. What is the cost of goods sold for the company?
-
Morenas Driving Schools 2015 statement of financial position showed non-current assets of 4.2 million. One year later, the 2016 statement of financial position showed non-current assets of 4.7...
-
A company has net income of 173,000, a profit margin of 8.6 per cent, and a trade receivables balance of 143,200. Assuming 75 per cent of sales are on credit, what is the companys days sales in...
-
For the year ending 2015, TJC plc has revenues of 1,027 million and total costs (excluding interest) of 817 million. Net interest expense was 29 million and depreciation was 12 million. What is the...
-
A financial ratio by itself tells us little about a company because financial ratios vary a great deal across industries. There are two basic methods for analysing financial ratios for a company:...
-
Find the equation of the conic section that passes through the points (0, 0), (0, - 1), (2, 0), (2, - 5), and (4, - 1).
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
An easy way to rationalize definitions of cycle performance is to think of them as: Thus, for an engine, thermal efficiency is =|W|/|Q H |; for a refrigerator, the coefficient of performance is =|Q C...
-
Devise a general scheme for analyzing four-step air-standard power cycles. Model each step of the cycle as a polytropic process described by PV = constant which implies that TP (1 ) = constant...
-
Air-standard power cycles are conventionally displayed on PV diagrams. An alternative is the PT diagram. Sketch air-standard cycles on PT diagrams for the following: (a) Carnot cycle (b) Otto cycle...
-
Consider the following information: State Probability ABC Inc.'s Return Boom 0.25 15% Normal 0.50 8% Slowdown 0.15 4% Recession 0.10 -3% i. 11. iii. What is the expected return? What is the variance?...
-
The mythical country of Quitar has just established a policy to give very generous in-kind benefits to the nation's poor, currently defined as those earning less than $10,000 a year. Several years...
-
Question 1 The external loadings imposed on a horizontal beam is shown in Figure Q1(a). The beam is pivoted at Point A. (a) (b) 100 N/m 50 N/m 20 N 40 cm 50 cm Figure Q1 70 cm Apply the principle of...

Study smarter with the SolutionInn App


