Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.70, initially at 70C and 100 kPa. Describe
Question:
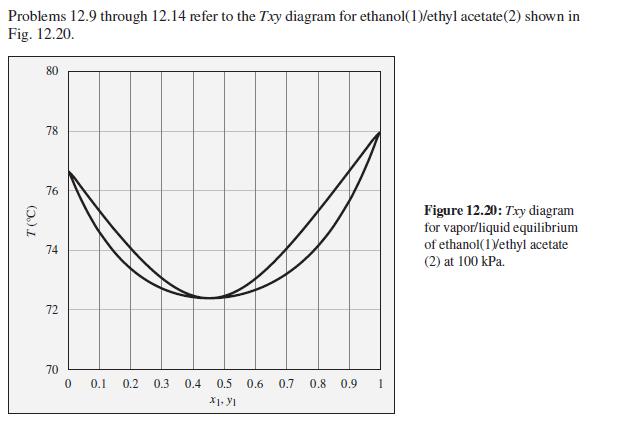
Consider an ethanol(1)/ethyl acetate(2) mixture with x1 = 0.70, initially at 70°C and 100 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually increased to 80°C.
Transcribed Image Text:
Problems 12.9 through 12.14 refer to the Txy diagram for ethanol(1)/ethyl acetate(2) shown in Fig. 12.20. 80 78 76 Figure 12.20: Txy diagram for vapor/liquid equilibrium of ethanol(1Vethyl acetate 74 (2) at 100 kPa. 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1 X1. YI (5.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
At the initial conditions the given ethanolethyl acetate mixture is a singlephase liquid As the temp...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Describe the evolution of business-to-business (B2B) e-commerce.
-
Describe the evolution of ERP systems.
-
Describe the evolution of American culinary arts.
-
How does MacKinnon analyze cases of sexual harassment where the woman complies with the sexual advance? She suggests that these cases are therefore not wrongful. She suggests that these cases are too...
-
1. Define pure economic rent. 2. Explain why the supply curve of land can be upwardsloping when there is a fixed quantity of land in the world. 3. Define artificial rent. 4. Define real rent. 5....
-
Over the years Best Buy and Circuit City have evolved into large corporations. Today it is difficult to imagine them as fledgling start-ups. Research each companys history online. Required 1. Which...
-
Prove that the order of convergence of the Crank-Nicolson finite difference method is \[O\left(\Delta x^{2}+\left(\frac{\Delta t}{2} ight)^{2} ight)\]
-
Duval Ford, LLC, sold a new Ford F-250 pick-up truck to David Sweat. Before taking delivery, Sweat ordered a lift kit to be installed on the truck by a Duval subcontractor. Sweat also replaced the...
-
You have a computer system where a customer wants the device to be changed from being a magnetic hard drive to an SSD using a M.2 interface. The desktop computer does not have a M.2 slot. Is what the...
-
Glentech Manufacturing is considering the purchase of an automated parts handler for the assembly and test area of its Phoenix, Arizona, plant. The handler will cost $ 250,000 to purchase plus $...
-
A mixture of ethanol and ethyl acetate is heated in a closed system at 100 kPa to a temperature of 77C, and two phases are observed to be present. What are the possible compositions of the liquid and...
-
Consider a closed vessel initially containing 1 mol of pure ethyl acetate at 70C and 86 kPa. Imagine that pure ethanol is slowly added at constant temperature and pressure until the vessel contains 1...
-
The following data compare the standard enthalpies and free energies of formation of some crystalline ionic substances and aqueous solutions of the substances: (a) Write the formation reaction for...
-
The Light Record Corporation has been on the calendar year since its inception five years ago. It wishes to change to an April 30 natural business year. For 2019, the calendar year of proposed...
-
Assume the same facts as the problem above except the premium is for coverage from August 1, 2019 to July 31, 2021. What is the largest deduction Wagner Co. can take in 2019? Data from in problem...
-
Lyle Inc. is a cash-basis, calendar-year taxpayer. In 2019, it changes to the accrual method of accounting, Its 2019 income computed under the accrual method is $75,000. The following information...
-
Do either of the transactions below qualify for installment reporting? If not, why not? a. Credit sales of dealer inventory b. Credit sales of property not held as inventory
-
As part of a study of young, UK-based black and minority business graduates career identities and aspirations, research participants were asked to talk about their future work-family balance. The...
-
Find the equations of the lines that pass through the following points: (a) (1, - 1), (2, 2) (b) (0, 1), (1, - 1)
-
Big Jim Company sponsored a picnic for employees and purchased a propane grill equipped with a standard-sized propane tank for the picnic. To make sure there was enough propane for all the cooking...
-
For a ternary solution at constant T and P, the composition dependence of molar property M is given by: M = x 1 M 1 + x 2 M 2 + x 3 M 3 + x 1 x 2 x 3 C where M 1 , M 2 , and M 3 are the values of M...
-
If the molar density of a binary mixture is given by the empirical expression: find the corresponding expressions for V 1 and V 2 p= ag + ajx1 +a2xf
-
Given below are values of G E /Jmol 1 , H E /Jmol 1 , and C E P / J mol 1 K 1 for some equimolar binary liquid mixtures at 298.15 K. Estimate values of G E , H E , and S E at 328.15 K for one of...
-
Saskatewan Can Company manufactures recyclable soft-drink cans. A unit of production is a case of 12 dozen cans. The following standards have been set by the production-engineering staff and the...
-
Assume that you hold 100 Vodacom shares which you intend to sell in a month's time. To protect yourself from future price risks, you then decided to construct a covered call strategy using a call...
-
The four people below have the following investments. Invested Amount Interest Rate Compounding Jerry Elaine $ 11,900 12% Quarterly 14,900 10 Semiannually 21,900 6 Annually 17,900 8 Annually George...

Study smarter with the SolutionInn App


