Consider an ethanol(1)/ethyl acetate(2) mixture with x 1 = 0.80, initially at 70C and 80 kPa. Describe
Question:
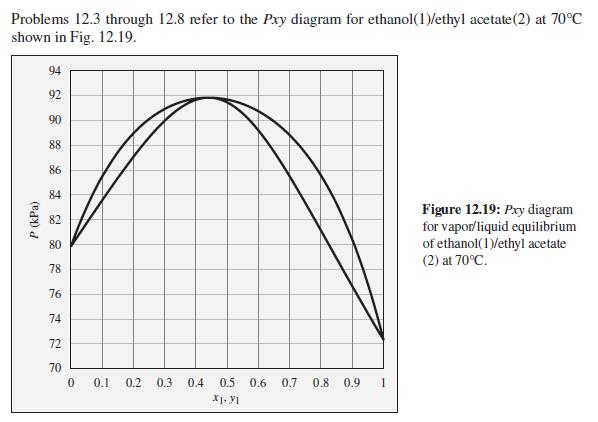
Consider an ethanol(1)/ethyl acetate(2) mixture with x1 = 0.80, initially at 70°C and 80 kPa. Describe the evolution of phases and phase compositions as the pressure is gradually increased to 100 kPa.
Transcribed Image Text:
Problems 12.3 through 12.8 refer to the Pxy diagram for ethanol(1)/ethyl acetate(2) at 70°C shown in Fig. 12.19. 94 92 90 88 86 84 Figure 12.19: Pry diagram for vapor/liquid equilibrium of ethanol(1)/ethyl acetate (2) at 70°C. 82 80 78 76 74 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 X1. y1 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
At 70C and 80 kPa the mixture is in a twophase region The compositions of the ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Describe the evolution of business-to-business (B2B) e-commerce.
-
Describe the evolution of ERP systems.
-
Describe the evolution of American culinary arts.
-
Scott incorporates his sole proprietorship as Superior Corporation and transfers its assets to Superior in exchange for all 100 shares of Superior stock and four $7,500 interest-bearing notes. The...
-
1. Describe the difference between the nominal interest rate and the real interest rate. 2. Explain why the real interest rate matters to borrowers and lenders.
-
Burton Companys fixed budget performance report for July follows. The $675,000 budgeted expenses include $634,500 variable expenses and $40,500 fixed expenses. Actual expenses include $52,500 fixed...
-
Use the implicit finite difference method to solve the heat conduction problem on the unit square: \[\begin{aligned}& \frac{\partial^{2} u}{\partial x^{2}}=\frac{\partial u}{\partial t} \\& u(x, 0)=x...
-
An industrial sewing machine uses ball bearings that are targeted to have a diameter of 0.75 inch. The lower and upper specification limits under which the ball bearings can operate are 0.74 inch and...
-
HomeWeb is a new real estate advertising company in Melbourne, Australia. The company has around 30 staff taking care of their main company website where real estate agents pay to post their...
-
The financial statements of JJ Ltd and KK Ltd for the year to 30 June 2018 are shown below: Statements of comprehensive income for the year to 30 June 2018. Statements of financial position as at 30...
-
Consider a closed vessel initially containing 1 mol of pure ethyl acetate at 70C and 86 kPa. Imagine that pure ethanol is slowly added at constant temperature and pressure until the vessel contains 1...
-
What is the composition of the azeotrope for the ethanol(1)/ethyl acetate(2) system? Would this be called a high-boiling or low-boiling azeotrope? Problems 12.3 through 12.8 refer to the Pxy diagram...
-
Since graduating from college five years ago, you have worked for a national chain of mens clothing stores. You have held several positions within the company and are currently manager of a local...
-
Steven Seller owned and managed a clothing store that was destroyed by a hurricane. He received an insurance award of $200,000 for the store, which had an adjusted basis of $120,000. A year later he...
-
Briefly define total deviation, unexplained deviation, and explained deviation.
-
Explain what we mean by the goodness of fit for a regression. Give two measures we can use to assess the goodness of fit.
-
Explain the difference between a population and a subpopulation.
-
What is causality? What is correlation? What is the relationship among casualty, correlation, and regression analysis?
-
Find the eigenvalues of the matrix where Ï = e2Ïi/3. 1 001 010
-
Refer to the Conservation Ecology (Dec. 2003) study of the causes of forest fragmentation, presented in Exercise 2.166 (p. 97). Recall that the researchers used advanced high-resolution satellite...
-
The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 52 kPa. What are the possible compositions of the liquid and vapor phases? To the Pxy diagram for...
-
The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 62 kPa. What are the possible compositions of the liquid and vapor phases? To the Pxy diagram for...
-
The following liquids, all at atmospheric pressure and 300 K, are mixed: 25 kg of pure water, 40 kg of pure sulfuric acid, and 75 kg of 25-wt-% sulfuric acid. (a) How much heat is liberated if mixing...
-
Question: d. ?During December, Ingrid Legal Services provided legal services, and the client prepaid $7,000. ?Ingrid Legal Services recorded this amount as Unearned Revenue. The job will take several...
-
Question: A company had the following purchases and sales during the month of November:\table[[Date,Activities,Units Acquired at Cost,Units Sold at Retail],[November 1,Beginning inventory,5 units @...
-
Question: Tania, Inc. issued a note payable with principal and interest due at the end of the term of the note. Details about the note payable are as follows:Issuance date: October 1, fear...

Study smarter with the SolutionInn App


