Question: Write the equilibrium constant expression for: (a) SiC14 (1) + 2 HO(g) (b) H(g) + CO(g) CO(g) + HO(1) (c) MnO(s) + 4H* (aq) +
Write the equilibrium constant expression for:
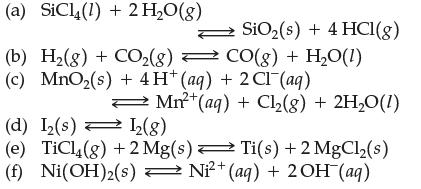
(a) SiC14 (1) + 2 HO(g) (b) H(g) + CO(g) CO(g) + HO(1) (c) MnO(s) + 4H* (aq) + 2 Cl(aq) SiO (s) + 4 HCl(g) Mn+ (aq) + Cl(g) + 2HO(1) (d) I(s) 1(8) (e) TiCl4(g) +2 Mg(s) Ti(s) + 2MgCl(s) 2+ (f) Ni(OH)(s) Ni+ (aq) + 2 OH (aq)
Step by Step Solution
★★★★★
3.20 Rating (161 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The equilibrium constant expressions for the reacti... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


