Use the periodic table to predict the ionic charge for each of the following nonmetal ions. (a)
Question:
Use the periodic table to predict the ionic charge for each of the following nonmetal ions.
(a) F ion
(b) Br ion
(c) S ion
(d) N ion.
Periodic Table
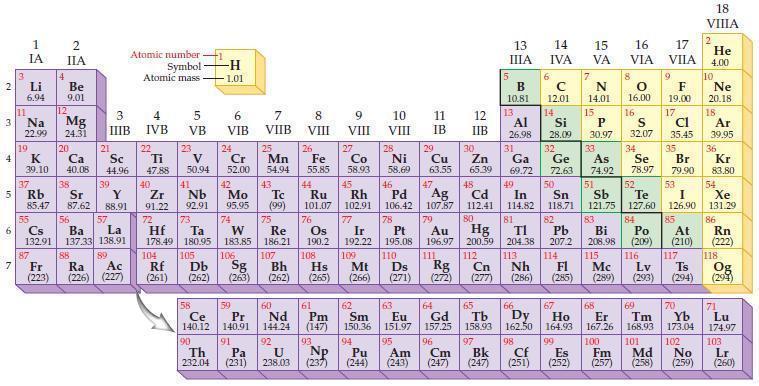
Transcribed Image Text:
2 3 4 5₁ 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca Sc 39.10 40.08 44.96 Mg 24.31 38 Rb Sr Y 85.47 87.62 88.91 56 57 Cs Ba La 132.91 137.33 138.91 20 21 88 3 IIIB 39 89 Atomic number Symbol - Ac Ra (226) (227) Atomic mass 4 IVB 22 Ti 47.88 40 5 VB 23 V 50.94 41 Zr Nb 91.22 92.91 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 108 9 VIII 61 Pm (147) 27 Bh Hs Mt (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB Pt 195.08 110 29 Cu 63.55 13 IIIA 12 IIB 5 B 10.81 13 17 16 VA VIA VIIA 8 6 C 12.01 14 Si 28.09 32 30 31 33 As Se Br Zn Ga Ge 65.39 69.72 72.63 74.92 78.97 79.90 Al 26.98 14 15 IVA 7 N 14.01 15 P 30.97 16.00 83 Bi 208.98 115 16 S 32.07 34 84 47 48 49 50 51 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 79 Au Hg 196.97 200.59 111 112 Rg Cn (271) (272) (277) (286) (285) (289) (293) (294) 81 82 TI Pb 204.38 207.2 113 114 Nh Fl Ds Mc Lv Ts Po (209) 9 116 F 19.00 Md (258) 17 Cl 35.45 35 53 I 126.90 85 At (210) 66 67 69 70 63 64 65 68 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 98 99 100 Cf Es Fm (251) (252) (257) 117 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To predict the ionic charges for nonmetal ions you can refer to the group number or the number of va...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Use the periodic table to predict an ionic charge for each of the following metal ions. (a) Na ion (b) Ba ion (c) Ga ion (d) Pb ion. Periodic Table 2 3 4 5 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55...
-
Use the periodic table to predict an ionic charge for each of the following metal ions. (a) Li ion (b) Mg ion (c) Al ion (d) Sn ion. Periodic Table 2 3 4 5 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55...
-
Use the periodic table to predict the ionic charge for each of the following nonmetal ions. (a) Cl ion (b) I ion (c) Se ion (d) P ion. Periodic Table 2 3 4 5 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al...
-
You are deciding between two mutually exclusive investment opportunities. Both require the same initial investment of $10 million. Investment A will generate $2 million per year (starting at the end...
-
Is a trustee appointed in Title 11 cases? In all Chapter 7 cases? Discuss.
-
A free particle of mass m is confined to a ring of circumference L such that (x+L) = (x). The unperturbed Hamiltonian is to which we add a perturbation (a) Show that the unperturbed states may be...
-
Two very long wires each carry a linear charge density \(\lambda\). They initially repel each other with a force \(F\). If the wires are immersed in distilled water, with what force do they repel...
-
The Sequel Theater Inc. was recently formed. It began operations in March 2014. The Sequel is unique in that it will show only triple features of sequential theme movies. On March 1, the ledger of...
-
Step 3: Testing Your Configuration Update 1. Restart the SSH service. [Your solution command here] 2. Exit the root account. [Your solution command here] 3. SSH to the target machine using your sysd...
-
Based on electronegativity trends in the periodic table, which of the following molecules is most polar: HF, HCl, HBr, HI?
-
Burning yellow sulfur powder produces sulfur dioxide, SO 2 . Sulfur dioxide is a colorless gas with a suffocating odor. It is used to kill insect larvae and produces the odor released when a match is...
-
If average productivity falls, will marginal cost necessarily rise? How about average cost?
-
In this question, you will analyze their Measures of Profitability: Although Rush Industries' gross profit margin is ( a . below or b . above ) its industry average, the company has a ( n ) ( a ....
-
Of all the price analysis methodologies that exist, which are the foundational methods. Longing & Shorting. Trading & Investing. Volume & sentiment. On-chain & Macroeconomic. Mean reversion &...
-
Let f(x1, x2)=-x-2x+x1x2 + 4x1 +5x2. (a) Compute Vf(x) and V2 f(x). (b) Find all local minima and maxima. (c) Check whether for -f is convex, and find out whether f has a global minimum or global...
-
Find two regression equations when it is given that X=68.2, Y=9.9, OY = 0.44 and r = 0.7.
-
3. Considering the following state diagram for a 3-bits counter: 000 001 010 011 111 110 101 100 (a) Design the circuit using T flip-flops. (b) Using the Verilog modules we wrote in the sequential...
-
Luke trades his baseball card collection for an automobile. The automobile is worth $11,000, and Luke assumes the $3,000 loan on the car. Luke has $3,500 invested in his baseball card collection....
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
Contribution margin , decision making Schmidt Men?s Clothing?s revenues and cost data for 2009 are: Contribution margin, decision making Schmidt Men?s Clothing?s re" alt = " Contribution margin ,...
-
Contribution margin , gross margin, and margin of safety Mirabella Cosmetics manufactures and sells a face cream to small ethnic stores in the greater New York area. It presents the monthly operating...
-
Uncertainty and expected costs. Dawmart Corp, an international retail giant, is considering implementing a new business to business (B2B) information system for processing purchase orders. The...
-
An Ice cream company has commissioned an ice sculpture for their end of year celebration. The ice sculpture is in the shape of a giant ice cream cone-the shape can be thought of as a cone with one...
-
1. A soft drink vendor at a popular beach resort analyzes his sales records and finds that if he seels x cans of pop in one day, his profit in dollars is given by the function P(x) = -0.012+3x-80....
-
1. Consider the following pseudocode. What does it produce? Set a = 0 Set b = 0 Set c = 1 Set d = 1 Report the value of d Repeat until a equals 10 Set d = b + c Set b = c Set c = d Add 1 to a Report...

Study smarter with the SolutionInn App


