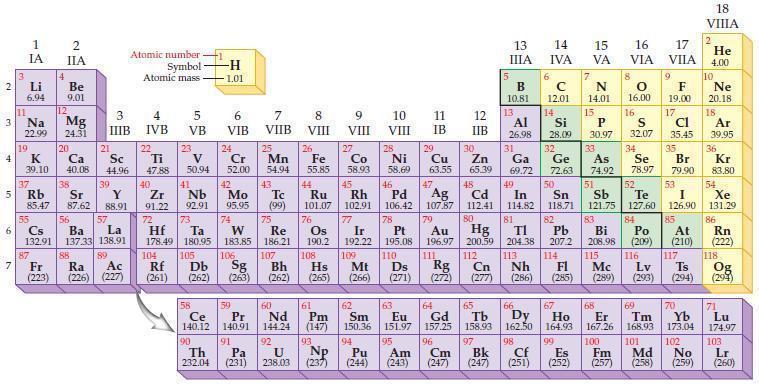
What is the general trend in electronegativity within a group of elements in the periodic table?
Question:
What is the general trend in electronegativity within a group of elements in the periodic table?
Periodic Table
Transcribed Image Text:
2 3 4 5₁ 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca Sc 39.10 40.08 44.96 Mg 24.31 38 Rb Sr Y 85.47 87.62 88.91 56 57 Cs Ba La 132.91 137.33 138.91 20 21 88 3 IIIB 39 89 Atomic number Symbol - Ac Ra (226) (227) Atomic mass 4 IVB 22 Ti 47.88 40 5 VB 23 V 50.94 41 Zr Nb 91.22 92.91 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 108 9 VIII 61 Pm (147) 27 Bh Hs Mt (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB Pt 195.08 110 29 Cu 63.55 13 IIIA 12 IIB 5 B 10.81 13 17 16 VA VIA VIIA 8 Al 26.98 6 C 12.01 14 Si 28.09 32 30 31 33 As Se Br Zn Ga Ge 65.39 69.72 72.63 74.92 78.97 79.90 14 15 IVA 7 N 14.01 15 P 30.97 16.00 83 Bi 208.98 115 16 S 32.07 34 84 47 48 49 50 51 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 79 Au Hg 196.97 200.59 111 112 Rg Cn (271) (272) (277) (286) (285) (289) (293) (294) 81 82 TI Pb 204.38 207.2 113 114 Nh Fl Ds Mc Lv Ts Po (209) 9 116 F 19.00 Md (258) 17 Cl 35.45 35 53 I 126.90 85 At (210) 66 67 69 70 63 64 65 68 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 98 99 100 Cf Es Fm (251) (252) (257) 117 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The general trend in electronegativity within a group of elements in the periodic table is that it d...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
What is the general trend in electronegativity within a period of elements in the periodic table? Periodic Table 2 3 4 5 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca...
-
a. Look at Figure 11.3. i. What is the general trend in the melting points going down Group 2? ii. Which element breaks the trend? b. Explain why the atoms in Group 2, as in any other group, get...
-
a. What is the general trend in first ionisation energies across Period 3? b. Explain why aluminium has a lower first ionisation energy than magnesium. c. Explain why sulfur has a lower first...
-
Jellio, Maker of Whimsical Furnishings, Considers Growth. Start by asking yourself, who is the customer for this product, not who is currently buying it. There is a difference. Then, where would they...
-
How does the accounting treatment of a nine-month note payable differ from the accounting treatment of a five-year note payable within a governmental fund? Why?
-
In assume that females have pulse rates that are normally distributed with a mean of 74.0 beats per minute and a standard deviation of 12.5 beats per minute (based on Data Set 1 Body Data in Appendix...
-
Write expressions for the magnitudes and directions of the electric fields measured in Problem 24. Data from Problem 24 A positively charged particle is at rest on the positive \(z\) axis in...
-
Direct Marketing Inc. (DMI) offers database marketing strategies to help companies increase their sales. DMIs basic package of services includes the design of a mailing piece (either a Direct Mailer...
-
4. The atomic numbers of elements, U, V, W and X are 13, 14, 16 and 17 respectively. Which one of the elements will react with oxygen to form an ionic compound? (Atomic number of oxygen is 8) X A. B....
-
Write the electron dot formula and draw the structural formula for each of the following polyatomic ions. (a) H 3 O + (b) OH (c) HS (d) CN .
-
Write the electron dot formula and draw the structural formula for each of the following polyatomic ions. (a) PO 4 3 (b) HPO 4 2 (c) PO 3 3 (d) HPO 3 2 .
-
Citalopram is used as an antidepressant. A biologist, Heywood U. Clonum, has argued that this compound could be hazardous because it could release toxic cyanide ( C N) and fluoride ions by...
-
Discuss how the Australian government and financial regulators have responded to the COVID-19 outbreak throughout 2020 to support the flow of credit to businesses.
-
'The fiscal and monetary authorities in the US have overreacted to the Covid-19 crisis; there is now excessive stimulus, both fiscal and monetary, in the US economy and this is bound to cause...
-
In this part, your goal is to derive the transfer function representation for the rotational mechanical system composed of two inertias Um and JL) and a damper (B) shown on the right. The input of...
-
If the risk rate of the return is 4%, the return on the market portfolio is 12%, the beta of the hotel norwood is 1.5, what is the expected rate of the return for hotel norwood?
-
10 cumecs of water is delivered to a 32 ha field for 4 hr. Calculate water application efficiency, if the depth of water stored in the root zone is 0.3 m.
-
Losses on exchanges must be deferred. A loss on an involuntary conversion is never deferred. In contrast, a loss on the sale of a principal residence is never recognized. Explain why losses on the...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Using the BAR Model given the following information, calculate the target cash balance using the BAT model: how do you interpret your answer? Annual interest rate 7% Fixed order cost $25 Total cash...
-
Opportunity versus Trading Costs White Whale Corporation has an average daily cash balance of $900. Total cash needed for the year is #37,000. The interest rate is 5 percent, and replenishing the...
-
Costs and the BAT Model Debit and credit bookkeepers needs a total of $7,000 in cash during the year for transactions and other purposes. Whenever cash runs low, it sells $450 in securities and...
-
You are considering the following two projects and can take only one. Your cost of capital is 10.5%. The cash flows for the two projects are as follows ($ million): Project A B a. What is the IRR of...
-
land value for 2022 was 110,00 and for 2023 is 104,000. the land was sold with a a gain of 7,400. Calculate amounts to enter in a statement of cash flows for 2023
-
Nance Network Consultants, Incorporated uses the percentage - of - completion method to account for its long - term contracts. It uses the cost - to - cost approach to measure progress. During the...

Study smarter with the SolutionInn App


