What is the general trend in electronegativity within a period of elements in the periodic table? Periodic
Question:
What is the general trend in electronegativity within a period of elements in the periodic table?
Periodic Table
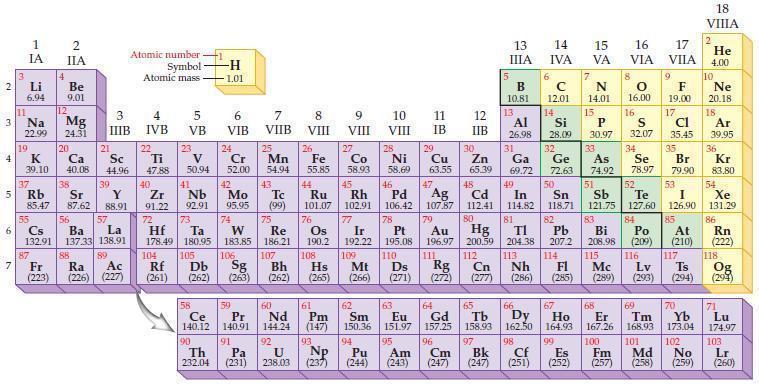
Transcribed Image Text:
2 3 4 5₁ 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca Sc 39.10 40.08 44.96 Mg 24.31 38 Rb Sr Y 85.47 87.62 88.91 56 57 Cs Ba La 132.91 137.33 138.91 20 21 88 3 IIIB 39 89 Atomic number Symbol - Ac Ra (226) (227) Atomic mass 4 IVB 22 Ti 47.88 40 5 VB 23 V 50.94 41 Zr Nb 91.22 92.91 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 108 9 VIII 61 Pm (147) 27 Bh Hs Mt (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB Pt 195.08 110 29 Cu 63.55 13 IIIA 12 IIB 5 B 10.81 13 17 16 VA VIA VIIA 8 Al 26.98 6 C 12.01 14 Si 28.09 32 30 31 33 As Se Br Zn Ga Ge 65.39 69.72 72.63 74.92 78.97 79.90 14 15 IVA 7 N 14.01 15 P 30.97 16.00 83 Bi 208.98 115 16 S 32.07 34 84 Po (209) 47 48 49 50 51 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 79 Au Hg 196.97 200.59 111 112 Rg Cn (271) (272) (277) (286) (285) (289) (293) (294) 81 82 TI Pb 204.38 207.2 113 114 Nh Fl Ds Mc Lv Ts 9 116 F 19.00 Md (258) 17 Cl 35.45 35 53 I 126.90 85 At (210) 66 67 69 70 63 64 65 68 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 98 99 100 Cf Es Fm (251) (252) (257) 117 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The general trend in electronegativity within a period horizontal row of elements in the perio...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
What is the general trend in electronegativity within a group of elements in the periodic table? Periodic Table 2 3 4 5 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca...
-
a. What is the general trend in first ionisation energies across Period 3? b. Explain why aluminium has a lower first ionisation energy than magnesium. c. Explain why sulfur has a lower first...
-
The accompanying graphs show the first ionization energies and electron affinities of the period 3 elements. Refer to the graphs to answer the questions that follow. a. Describe the general trend in...
-
Analyzing Revenues and Expenses and Completing an Income Statement Neighborhood Realty, Incorporated, has been operating for three years and is owned by three investors. S. Bhojraj owns 60 percent of...
-
What does measurement focus mean? What two focuses are used in governmental accounting? Which fund types use each?
-
Between 2.5 minutes and 4.5 minutes In refer to the continuous uniform distribution depicted in Figure 6-2 and described in Example 1. Assume that a passenger is randomly selected, and find the...
-
If two charged particles \(M\) and \(S\) are at rest relative to each other, there is no magnetic force between them. Suppose instead that particle \(M\) is moving relative to particle \(S\) while...
-
Joey Department Store's employees are paid on the 6th and 22rd of each month for the period ending the last day of the previous month and the 15th of the current month, respectively. An analysis of...
-
22 22 The atomic number of an element T is 10. T shows similar properties to an element with atomic number 23. 24. A. 14 B. 16 C. 18 d. 20 The electron configurations of elements W, X, Y and Z are...
-
Write the electron dot formula and draw the structural formula for each of the following polyatomic ions. (a) PO 4 3 (b) HPO 4 2 (c) PO 3 3 (d) HPO 3 2 .
-
Write the electron dot formula and draw the structural formula for each of the following polyatomic ions. (a) SO 4 2 (b) HSO 4 (c) SO 3 2 (d) HSO 3 .
-
Explain the three-factor model of job design.
-
Recalculate the intrinsic value of Toyota shares using the free cash flow model of Spreadsheet(above) under each of the following assumptions. Treat each scenario independently. a. Toyota's P/E ratio...
-
% % 3% 3% 0% 1% 7% 3% 6% 1% 12% 92% 02% 15% Calculate the expected return and standard deviation for each asset individually using the data in the table to the left. StockB StockC Expected Return...
-
Question 25 1 points You plan to invest $100 for two years with a building society. The building society pays 6% per annum compounded monthly. The effective annual rate of interest earned on the...
-
Question: Should we use the recommended 4% discount rate? B-Well Health Mart has a capital structure consisting of 30% debt and 70% equity. The debt consists of loans from the Long Island Bank with...
-
Find the implied volatility (to 2 decimals, for example, o = 8.23%) of a Put option with a time to expiration of 11 months and a price of $6.13 The stock is currently trading at $47. The riskless...
-
How long does a taxpayer who suffers an involuntary conversion of an asset have to replace the asset to qualify for non-recognition? Explain.
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
Interpreting Miller-Orr based on the Miller-Orr model describe what will happen to the lower limit, the upper limit, and the spread (the distance between the two) if the variation in net cash flow...
-
Using Miller-Orr the variance of the daily cash flows for the Pele Bicycle Shop is $840,000. The opportunity cost to the firm of holding cash is 7 percent per year. What should the target cash level...
-
Using bat all night corporations has determined that its target cash balance if it uses the BAT model is $2,600. The total cash needed for the year is $23,000, and the order cost is $10. What...
-
What are the advanced methodologies and best practices for database indexing, considering factors such as data distribution, query patterns, and storage engine characteristics, to maximize query...
-
How does the evolving landscape of database technologies, encompassing both relational and non-relational paradigms, address the challenges posed by the proliferation of big data, streaming data, and...
-
6. Find a closed form for the generating function for the sequence {an}, where a) an = -1 for all n = 0, 1, 2,.... b) an = 2" for n = 1, 2, 3, 4, ... and ao = 0. c) an=n-1 for n = 0, 1, 2, .... d) an...

Study smarter with the SolutionInn App


