(a) As shown in Fig. P11.78(a), 1,5-cyclooctadiene undergoes an electrophilic addition with SCl2 to give compound A....
Question:
(a) As shown in Fig. P11.78(a), 1,5-cyclooctadiene undergoes an electrophilic addition with SCl2 to give compound A. (Notice the conformation of A, also shown.) Provide a curved-arrow mechanism for this transformation that accounts for the stereochemistry.
(b) Suggest a mechanism that accounts for the reaction of A shown in Fig. P11.78(b).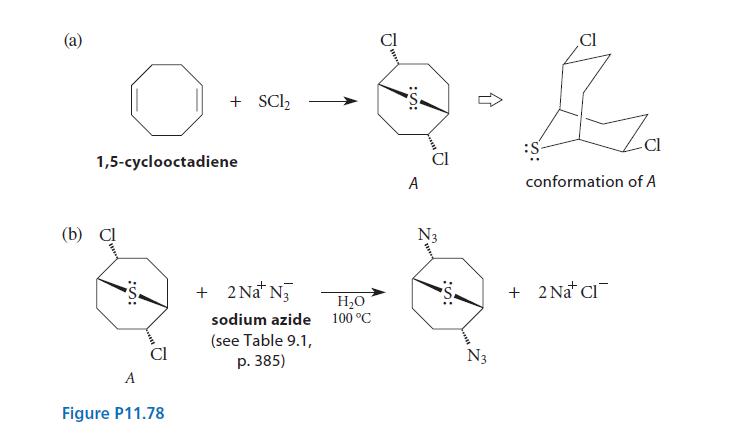
Transcribed Image Text:
(a) 1,5-cyclooctadiene (b) Cl :S: + SC1₂ A Figure P11.78 + 2Na+ N₂ sodium azide (see Table 9.1, p. 385) H₂O 100 °C :S: A N3 :S: Cl Za conformation of A :S + 2 Na Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a b An episulfonium ion analogous to a bromonium ion X is formed at one double bond r...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In what ways is this issue similar to and different from controversies about using illegal aliens to perform low-wage jobs inside the U.S.?
-
The S N 2 reaction between a Grignard reagent and an epoxide works reasonably well when the epoxide is ethylene oxide. However, when the epoxide is substituted with groups that provide steric...
-
For the same steam power plant as shown in Fig P6.99 and Problem 6.167E determine the rate of heat transfer in the economizer which is a low temperature heat exchanger and the steam generator....
-
Managers should be supported through process transitions by ___________. Question 12 options: encouragement (coaching and mentoring) and assistance (necessary resource) structured approach with a...
-
Assume that Key Co. purchased 1,000 units of merchandise in its first year of operations for $25 per unit. The company sold 850 units for $40. What is the amount of cost of goods sold using FIFO?...
-
Petgirl Company earned $25,000 of revenues and incurred $12,950 worth of expenses during the period. Petgirl also declared and paid dividends of $1,500 to its shareholders. What was net income for...
-
Oncology Tech is a manufacturer of cancer therapy devices called compensators. CMS was a company that sold cancer treatment planning software for use in purchasing cancer treatment devices. In 2006,...
-
In its annual report to stockholders, Hakobe Inc. presents a condensed balance sheet with detailed data provided in supplementary schedules. 1. From the adjusted trial balance of Hakobe, prepare the...
-
1.Libby just expanded her restaurant. She projects revenue will reach $35,000 for the new restaurant in the first year and increase by 25% over the next three years. Expenses are 75% of sales. The...
-
Calculate the frequency of (a) Infrared light with = 9 * 10 6 m (b) Blue light with = 4800
-
One of the reactions given in Fig. P11.76 is about 2000 times faster in pure water than it is in pure ethanol. Another is about 20,000 times faster in pure ethanol than it is in pure water. The rate...
-
Discuss several factors that were completely ignored in our analysis of the gasoline mileage problem.
-
What is a structured note?
-
a. What is Euro commercial paper? b. How does it differ from U.S. commercial paper?
-
What is meant by a credit or quality spread?
-
What role do rating companies play in financial markets?
-
What are the different forms of credit risk?
-
Herbert and Geraldine have a taxable income of $28,000 before considering the gain they realize on the sale of 500 shares of Olebolla Corporation common stock for $26 per share. Herbert had acquired...
-
A statistical study shows that the fraction of television sets of a certain brand that are still in service after x years is given by f (x) = e-0.15x. (a) What fraction of the sets are still in...
-
Fill in the missing reagents a?c in the following scheme: NH2 CHCH3 CH=CH2 CCH3 b, c CHCH2NCH3 CH CH-CH2
-
Although pyrrole is a much weaker base than most other amines, it is a much stronger acid (pK a 15 for the pyrrole versus 35 for diethyl amine). The NH proton is readily abstracted by base to yield...
-
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity, and explain yourordering. NH2 Histamine H.
-
KADS, Inc. is planning to spend $300,000 on a machine to produce the new game. The machine has an expected life of three years, a $100,000 estimated resale value at the end of year 3, and will be...
-
Suppose your firm is considering investing in a project with the cash flows shown as follows, that the required rate of return on projects of this risk class is 10 percent, and that the maximum...
-
An App for personal finances offers a subscription service. Price is $10 per month. Variable costs are 0.1$ per month and client. Marketing spendings are $5 per year and client. Companys attrition...

Study smarter with the SolutionInn App


