Consider the reaction of a methyl radical (CH3) with the p bond of an alkene: The relative
Question:
Consider the reaction of a methyl radical (·CH3) with the p bond of an alkene: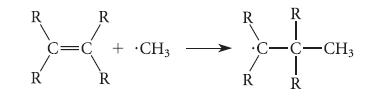
The relative rates of the reaction shown in Fig. P5.50 were determined for various alkenes.
(a) Draw the free-radical product of the reaction in each case and explain.
(b) Explain the order of the relative rates.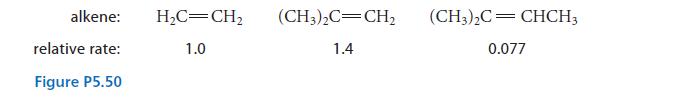
Transcribed Image Text:
R R T Hoa C=C + CH3 R R R R R R •C-C-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
a The methyl radical reacts at the carbon of the double bond with fewer substituents because this mode of reaction gives the more substituted radical ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction of a methyl ketone with iodine solution in the presence of aqueous sodium hydroxide is shown in the equation below: RCOCH3() + 3l2(aq) + 4NaOH(aq) RCOONa(aq) + CHl3(s) + 3Nal(aq) +...
-
Namib Mills, has a fixed total capital of $10,000,000, which is made up of 20 percent debt and 80 percent equity. The firm has 100,000 outstanding ordinary shares and no preference shares. Although...
-
Suppose an industry has the following data: TC=q-5q+100 a P=55-2Q If the industry is competitive; Calculate price, production quantity and profit. b. If there is a firm in the industry; Calculate...
-
Think about the impact that e-commerce and more flexible networks of organisations has had on the way international business is conducted. How has it created new complexities in the relationships...
-
H. Banks Company would like to design, produce, and sell versatile toasters for the home kitchen market. The toaster will have four slots that adjust in thickness to accommodate both slim slices of...
-
The December 31, 2011, year-end inventory balance of the Raymond Corporation is $210,000. You have been asked to review the following transactions to determine if they have been correctly recorded....
-
What the procedures are for a third-party stakeholder to deposit funds with the court?
-
Analyze the following independent situations. a. Sophia, Inc. is being sued by a former employee. Sophia believes that there is a remote chance that the employee will win. The employee is suing...
-
Required information On January 1st, 2024, Jurassic Pork, a food truck specializing in smoked ribs and pulled pork, is purchasing a new food truck for its fleet by signing a note payable which...
-
Equations 5.25ac on p. 193 show the formation of trialkylboranes from alkenes and BH 3 . In the reaction of 2,3-dimethyl-2-butene with BH 3 , only two equivalents of the alkene react, even with a...
-
Using the curved-arrow or fishhook notation, as appropriate, suggest mechanisms for each of the reactions given in Fig. P5.49. (a) HC (b) H3C C-CH CHCH=CH HC CH T T HO OH C#yan + Br H3O+ HC + CI-S-CI...
-
The publicity is widespread about MetLifes misdeeds. Debate how you would react. One position is to defend your company and rationalize what happened and downplay any ill-effects. The other position...
-
NYPD overwhelmingly stopped and still does stop mostly African-Americans and Hispanics in high-crime areas for no reason and would search them for weapons. Some who support the stop and frisk policy...
-
How and why do leaders personalities and organizational culture affect their performance? Are there practices to leverage organizational culture for a competitive advantage?
-
Analyze the economic feasibility of utilizing prefabricated building components (pre-fabs) for a high-rise office development in a developing nation, considering construction time constraints, local...
-
Ruby Tyler, a member of the United Pentecostal Church, was employed by a school district as an interpreter and tutor for deaf students. In this capacity, she worked at an elementary school for one...
-
(COVID-19 provisions) Lamden Company paid its employee Trudy, wages of $61,500 in 2021. Of this amount, $2,400 was allocated to sick pay for two weeks due to Trudy's spouse contracting COVID-19 and...
-
Identify the basic operations of relational algebra. What are they used for?
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
Draw an energy level diagram for the excited state of H2. Is there still a bond between the hydrogens?
-
The following code tries to delete a SLL. Test the code in Visual Studio Code and explain what you observed with detail. void cleanup(Node*& head) { Node current = head; while(current != nullptr) {...
-
Explain how do you cite the Donatos case study found in the textbook ?
-
Instructions: choose a recent article (i.e. published no earlier than January 2021) from either a newspaper, journal, magazine or internet. The article itself, or your review of the article, must...

Study smarter with the SolutionInn App


